
In 1976, a team of chemists in the United Kingdom was developing new insecticides by modifying sugars with chlorine (Cl2), phosgene (Cl2CO), and other toxic gases. One young member of the team misunderstood his verbal instructions to “test” a new molecule. He thought he had been told to “taste” it. Luckily for him, the molecule was not toxic, but it was very sweet. It became the food additive sucralose.
Sucralose has three chlorine atoms substituted for three hydroxyl groups of sucrose (table sugar):
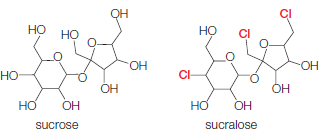
The altered sugar binds so strongly to the sweet-taste receptors on the tongue that the human brain perceives it as 600 times sweeter than sucrose. Sucralose was originally marketed as an artificial sweetener called Splenda®, but it is now available under several other brand names.
Researchers investigated whether the body recognizes sucralose as a carbohydrate by feeding sucralose labeled with 14C to volunteers. Analysis of the radioactive molecules in the volunteers’ urine and feces showed that 92.8 percent of the sucralose passed through the body without being altered.
Some people are worried that the chlorine atoms impart toxicity to sucralose. How would you respond to that concern?
Trending nowThis is a popular solution!

Chapter 3 Solutions
Biology: The Unity and Diversity of Life (MindTap Course List)
Additional Science Textbook Solutions
Loose Leaf For Integrated Principles Of Zoology
Campbell Biology (10th Edition)
Human Physiology
Campbell Biology: Concepts & Connections (8th Edition)
Laboratory Experiments in Microbiology (11th Edition)
- Figure 3.5 What kind of sugars are these, aldose or ketose?arrow_forwardAfter the removal of all sugar units from a ganglioside, what molecule remains? A. sphingosine B. a ceramide C. a glycerol D. a phosphatearrow_forwardUsing a maximum of 300 words, choose a health care product that is present in yourhouse. Choose one property of this product. Since aproduct is a a bunch of molecules interacting with eachother, how does molecular interaction leads to theproperty that you have identified?arrow_forward
- Which one of the following statements about the molecular structure of phosphatidylcholine is TRUE? a. It contains an isoprene chain. b. It contains a polar hydrophobic head group. c. It contains at least five alkene double bonds. d. It contains a phosphodiester bond. e. It contains a glycosidic linkage.arrow_forwardA multinational company outside india tried to sell new varieties of turmeric without proper patent rights.what is such an act referred to?arrow_forwardWhich of the following statements is FALSE? A. saccharin is 300 times sweeter than saccharose B. D-glucose is slightly salty and L-glucose is sweet. C. a-D-mannose is sweet and B-D-mannose is bitter D. The stereochemistry of flavor-producing agents plays an important role in the specific sensation of flavor.arrow_forward
- Many consumer products are almost identical in chemical composition but are sold under different brand names and at varying prices. Should some medical drugs be identified by their chemical (generic) names or trade names? Why?arrow_forwardChemistry Ninhydrin is used to turn amino acids in fingerprints purple, make a good way to stain fingerprints. The reaction of ninhydrin with amino acids is different for proline than the other amino acids. For 19 amino acids including glycine, it takes two ninhydrin molecules to react with the amino acid to produce the purple pigment products. For proline, only one ninhydrin molecule reacts and the color of the product is not normally purple. Can someone help me solve problems a-c? Thank you! Will thumbs up if correct!arrow_forwardAnthocyanins are another type of plant pigment that are purple or red. Say you run a chromatography experiment using the same setup as you did in this lab, and you find that anthocyanins do not move from the line of origin. Assuming the solvent traveled the same distance as in your experiment (D in Table 1 above), what is the Rf value of anthocyanin?arrow_forward
- Expansins are activated by acidic conditions that modify the Select one: a. Hydrogen bonds between pectin and cellulose b. Hydrogen bonds between pectin and glycoproteins c. Hydrogen bonds between cutin and cellulose d. Hydrogen bonds between lignin and cellulosearrow_forwardWhat would happen to a normal chemical process in your cells if the shape of the enzyme controlling that process was changed by an environmental toxin?arrow_forwardWhat structures are found in sterol molecules Select one: a. Waxes b. Double helixes c. Molecular rings d. Proteinsarrow_forward
 Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781337408332Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781337408332Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
 Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax
Biology 2eBiologyISBN:9781947172517Author:Matthew Douglas, Jung Choi, Mary Ann ClarkPublisher:OpenStax



