
Concept explainers
(a)
Interpretation:
To arrange the given covalent bonds within each set in the order of increasing polarity.
Concept Introduction:
When two atoms are bonded, because of the electronegativity difference between them, partial charge arises.
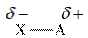
Thus, polarity results from the uneven partial charge difference. The difference in bonds collectively results in polarity of the molecule.
(b)
Interpretation:
To arrange the given covalent bonds within each set in the order of increasing polarity.
Concept Introduction:
When two atoms are bonded, because of the electronegativity difference between them, partial charge arises.
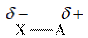
Thus, polarity results from the uneven partial charge difference. The difference in bonds collectively results in polarity of the molecule.
(c)
Interpretation:
To arrange the given covalent bonds within each set in the order of increasing polarity.
Concept Introduction:
When two atoms are bonded, because of the electronegativity difference between them, partial charge arises.
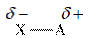
Thus, polarity results from the uneven partial charge difference. The difference in bonds collectively results in polarity of the molecule.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Introduction to General, Organic and Biochemistry
- 3-109 Until several years ago, the two chlorofluorocarbons (CFCs) most widely used as heat transfer media in refrigeration systems were Freon-li (trichloro fluoromethane, CC13F) and Freon-12 (dichiorodi fluoromethane, CCl2F2). Draw a three-dimensional representation of each molecule and indicate the Direction of it.s polarity.arrow_forward3-106 Consider the structure of Penicillin G shown below, an antibiotic used to treat bacterial infections caused by gram-positive organisms, derived from Penicillium fungi: (a) Identify the various types of geometries present in each central atom using VSEPR theory. (b) Determine the various relative bond angles associated with each central atom using VSEPR theory (c) Which is the most poiar bond in Penicillin G? (d) Would you predict Penicillin G to be polar or nonpolar?arrow_forward3-31 Why does electronegativity generally increase going from left to right across a row of the Periodic Table?arrow_forward
- 3-25 Why are carbon and silicon reluctant to form ionic bonds?arrow_forward3-67 Why does nitrogen have three bonds and one unshared pair of electrons in covalent compounds?arrow_forward3-68 Draw a Lewis structure of a covalent compound in which nitrogen has: (a) Three single bonds and one unshared pair of electrons (b) One single bond, one double bond, and one unshared pair of electrons (c) One triple bond and one unshared pair of electronsarrow_forward
- 3-107 Ephedrine, a molecule at one time found in the dietary supplement ephedra, has been linked to adverse health reactions, such as heart attacks, strokes, and heart palpitations. The use of ephedra in dietary supplements is now banned by the FDA. (a) Which is the most polar bond in ephedra? (b) Would you predict ephedra to be polar or nonpolar?arrow_forward3-70 Draw a Lewis structure of a covalent compound in which oxygen has: (a) Two single bonds and two unshared pairs of electrons (b) One double bond and two unshared pairs of electronsarrow_forward3-105 Consider the structure of Vitamin E shown below, which is found most abundantly in wheat germ oil, sunflower, and safflower oils: (a) Identify the various types of geometries present in each central atom using VSEPR theory. (b) Determine the various relative bond angles as sociated with each central atom using VSEPR theory. (c) Which is the most polar bond in Vitamin E? (d) Would you predict Vitamin E to be polar or nonpolar?arrow_forward
- 3-87 Consider the molecule boron trffluoride, BF3. (a) Write a Lewis structure for BF3. (b) Predict the FBF bond angles using the VSEPR model. (c) Does BF3 have polar bonds? Is it a polar molecule?arrow_forward3-119 Perchloroethylene, which is a liquid at room temperature, is one of the most widely used solvents for commercial dry cleaning. It is sold for this purpose under several trade names, including Perciene®. Does this molecule have polar bonds? Is it a polar molecule? Does it have a dipole?arrow_forward3-64 Acetylene (C2H2), hydrogen cyanide (HCN), and nitrogen (N2) each contain a triple bond. Draw a Lewis structure for each molecule. Which of these are polar molecules, and which are nonpolar molecules?arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
