
Concept explainers
(a)
Interpretation: The reaction of
Concept introduction: An atom or a group of atoms that shows characteristic physical and chemical properties are collectively known as
According to Bronsted-Lowry theory, the species that can easily accept the proton is known as base and the species that can easily donate the proton is known as acid. The reaction of an acid with a base always leads to the formation of conjugate acid and base.
Answer to Problem 3.1P
The reaction of
Explanation of Solution
The reaction of
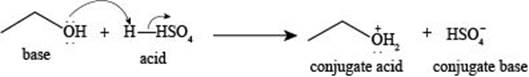
Figure 1
The reaction of ethanol
The reaction of
The reaction of ethane
The reaction of
(b)
Interpretation: The reaction of
Concept introduction: An atom or a group of atoms that shows characteristic physical and chemical properties are collectively known as functional groups. The functional group is the most reactive part present in the molecule. The main functional groups are
According to Bronsted-Lowry theory, the species that easily tends to accept the proton is known as base and the species that easily donate the proton is known as acid. The reaction of an acid with a base always leads to the formation of conjugate acid and base.
Answer to Problem 3.1P
The reaction of
Explanation of Solution
The reaction of
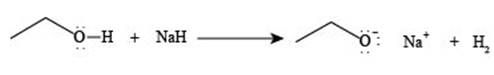
Figure 2
The reaction of ethanol
The reaction of
The reaction of ethane
The reaction of
Want to see more full solutions like this?
Chapter 3 Solutions
ORGANIC CHEMISTRY >ACCESS CODE<
- Draw the products formed when ethylene oxide is treated with following reagent. [1] −OH; [2] H2Oarrow_forwardDraw the product formed when cyclohexane carbaldehyde is reacted with NaOH and H2O.arrow_forwardWhat products are formed when benzoic acid (C 6H 5COOH) is treated with each base: (a) NaOH; (b) Na 2CO 3; (c) NaHCO 3?arrow_forward
- What ester is formed when each carboxylic acid is treated with ethanol (CH 3CH 2OH) in the presence of H 2SO 4?arrow_forward6. Aldehydes are characterized by reactions: A) Nucleophilic addition of amines B) Nucleophilic addition of water C) Nucleophilic addition of alcohols D) Polymerizationarrow_forwardDraw the products a. Pentyl butanoate reacts with water and NaOH b. Acetic anhydride reacts with ethanol c. 3-hexanone reacts with two equivalents of isopropyl alcohol d. Hexanal reacts with NaBH4 and CH3OHarrow_forward
- 1. Carboxylic acid reacts with an alcohol to form: A. Ester and Water B. Ester C. Water D. No reaction 2. The general formula for Carboxylic acids: A. RCOOH B. RCOOR C. RCOR D. RCOH 3. General formula of phenols: * A. ROH B. Ar-OH C. R-SH D. RCOHarrow_forwardDraw the products formed when CH3CH2C=C Na+ reacts with following compound. propene oxide followed by H2Oarrow_forwardMaltose is a carbohydrate present in malt, the liquid obtained from barley and other grains. Although maltose has numerous functional groups, its reactions are explained by the same principles we have already encountered.a. Label the acetal and hemiacetal carbons.b. What products are formed when maltose is treated with each of these reagents: [1] H3O+; [2] CH3OH and HCl; [3] excess NaH, then excess CH3I?c. Draw the products formed when the compound formed in Reaction [3] of part (b) is treated with aqueous acid.The reactions in parts (b) and (c) are used to determine structural features of carbohydrates like maltose.arrow_forward
- 8. What type of reaction takes place when CO2 + H2O and energy are obtained from 3-methyl butyne?a) Hydrogenationb) Halogenationc) Partial oxidationd) Total oxidationarrow_forwardIf a carboxylic acid is dissolved in isotopically labeled methanol (CH3 18OH) and an acid catalyst is added, where will the label reside in the product?arrow_forwardWhat is the structure of C4H6 that can react with NaNH2 and CH3I? What is the product? What reagents are needed to turn this Product into Z-2-pentene?arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning



