
Concept explainers
(a)
Interpretation:
Concept Introduction:
Atoms are made up of even smaller particles. These particles are very small and these are all the building blocks of atoms and they are known as subatomic particles. Protons, electrons, and neutrons are the subatomic particles that are found in atom. Electrons possess a negative electrical charge. Protons possess a positive electrical charge. Neutrons possess no charge and they are neutral.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
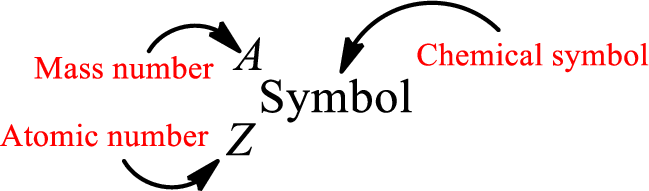
An element is a pure substance that cannot be broken by ordinary
(a)
Explanation of Solution
For
The atomic number is given as 18. The element is found to be argon. Mass number given for argon is given as 40. The number of protons present in it is also 18 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 22.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 40.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 58.
For
The atomic number is given as 20. The element is found to be calcium. Mass number given for calcium is given as 40. The number of protons present in it is also 20 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 20.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 40.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 60.
On comparing both the pair of atoms it is found that they contain same number of nucleons only.
(b)
Interpretation:
Concept Introduction:
Atoms are made up of even smaller particles. These particles are very small and these are all the building blocks of atoms and they are known as subatomic particles. Protons, electrons, and neutrons are the subatomic particles that are found in atom. Electrons possess a negative electrical charge. Protons possess a positive electrical charge. Neutrons possess no charge and they are neutral.
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
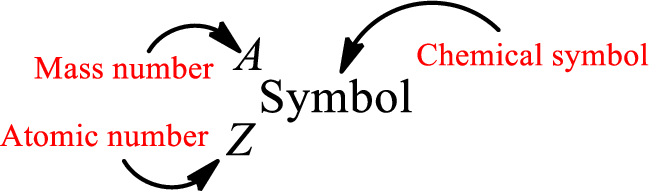
An element is a pure substance that cannot be broken by ordinary chemical reactions into simpler substances. All the atoms in an element will have the same atomic number. The electrons only take part in the chemical reaction while the nucleus does not. Hence, the atomic number (number or protons) does not change and it characterizes an atom.
(b)
Explanation of Solution
For
The atomic number is given as 7. The element is found to be nitrogen. Mass number given for nitrogen is given as 14. The number of protons present in it is also 7 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 7.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 14.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 21.
For
The atomic number is given as 7. The element is found to be nitrogen. Mass number given for nitrogen is given as 15. The number of protons present in it is also 7 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 8.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 15.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 22.
On comparing both the pair of atoms it is found that they contain same number of protons.
(c)
Interpretation:
Concept Introduction:
Atoms are made up of even smaller particles. These particles are very small and these are all the building blocks of atoms and they are known as subatomic particles. Protons, electrons, and neutrons are the subatomic particles that are found in atom. Electrons possess a negative electrical charge. Protons possess a positive electrical charge. Neutrons possess no charge and they are neutral.
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
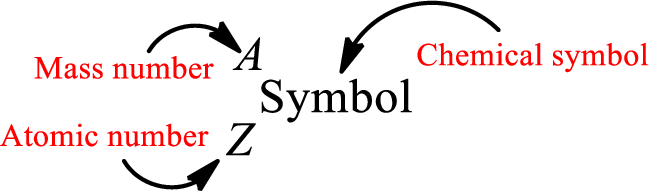
An element is a pure substance that cannot be broken by ordinary chemical reactions into simpler substances. All the atoms in an element will have the same atomic number. The electrons only take part in the chemical reaction while the nucleus does not. Hence, the atomic number (number or protons) does not change and it characterizes an atom.
(c)
Explanation of Solution
For
The atomic number is given as 6. The element is found to be carbon. Mass number given for carbon is given as 14. The number of protons present in it is also 6 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 8.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 14.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 22.
For
The atomic number is given as 7. The element is found to be nitrogen. Mass number given for nitrogen is given as 15. The number of protons present in it is also 7 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 8.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 15.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 22.
On comparing both the pair of atoms it is found that they contain same number of neutrons.
(d)
Interpretation:
Concept Introduction:
Atoms are made up of even smaller particles. These particles are very small and these are all the building blocks of atoms and they are known as subatomic particles. Protons, electrons, and neutrons are the subatomic particles that are found in atom. Electrons possess a negative electrical charge. Protons possess a positive electrical charge. Neutrons possess no charge and they are neutral.
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
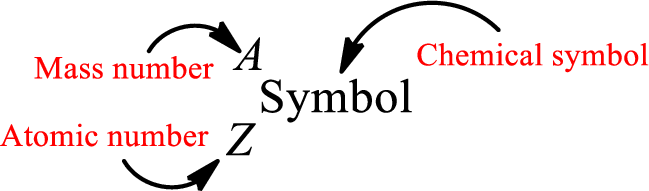
An element is a pure substance that cannot be broken by ordinary chemical reactions into simpler substances. All the atoms in an element will have the same atomic number. The electrons only take part in the chemical reaction while the nucleus does not. Hence, the atomic number (number or protons) does not change and it characterizes an atom.
(d)
Explanation of Solution
For
The atomic number is given as 18. The element is found to be argon. Mass number given for argon is given as 38. The number of protons present in it is also 18 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 20.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 38.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 56.
For
The atomic number is given as 19. The element is found to be potassium. Mass number given for potassium is given as 39. The number of protons present in it is also 19 as atomic number is the number of protons or electrons. Mass number of the sum of protons and neutrons present in the atom.
The number of neutrons can be identified by finding the difference between mass number and atomic number. This gives the number of neutrons to be 20.
The total number of nucleons is same as that of the mass number. This means the total number of nucleons is 39.
The total number of subatomic particle present in the given atom is the sum of mass number and atomic number. This means the total number of subatomic particle is 58.
On comparing both the pair of atoms it is found that they contain same number of neutrons.
Want to see more full solutions like this?
Chapter 3 Solutions
General, Organic, and Biological Chemistry
- 2.10 Which isotope in each pair contains more neutrons? (A) 35Cl or 33S, (b) 19F or 19Ne, (c) 63Cu or 65Zn, (d) 126I or 127Tearrow_forwardAn average atomic nucleus has a diameter of about m.arrow_forwardIt is good practice to actively read the textbook and to try to verify claims that are made when you can. The following claim is made in your textbook: “. . . if the nucleus were the size of a grape, the electrons would be about 1 mile away on average.” Provide mathematical support for this statement.arrow_forward
- How did the charge distribution in the plum pudding model affect alpha particles passing through an atom?arrow_forward2.86 For some uses, the relative abundance of isotopes must be manipulated. For example, a medical technique called boron neutron capture therapy needs a higher fraction of 10B than occurs naturally to achieve its best efficiency. What would happen to the atomic weight of a sample of boron that had been enriched in 10B? Explain your answer in terms of the concept of a weighted average.arrow_forwardWhile traveling to a distant universe, you discover the hypothetical element X. You obtain a representative sample of the element and discover that it is made up of two isotopes, X-23 and X-25. To help your science team calculate the atomic weight of the substance, you send the following drawing of your sample with your report. In the report, you also inform the science team that the brown atoms are X-23, which have an isotopic mass of 23.02 amu, and the green atoms are X-25, which have an isotopic mass of 25.147 amu. What is the atomic weight of element X?arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co





