
Concept explainers
(a)
Interpretation:
The number of protons, neutrons and electrons present in the atom of
Concept Introduction:
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
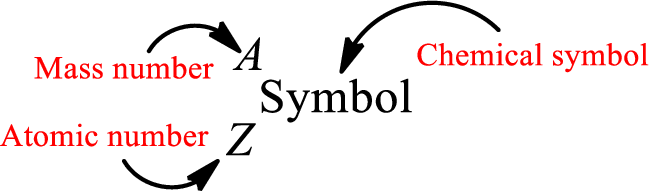
An element is a pure substance that cannot be broken by ordinary
(a)
Explanation of Solution
Given atom is
Atomic number is given as 50 and mass number is given as 118.
Therefore, the number of protons is 50, number of electrons is 50, and number of neutrons is 68.
The number of protons, electrons and neutrons present in the atom is given.
(b)
Interpretation:
The number of protons, neutrons and electrons present in the atom of
Concept Introduction:
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
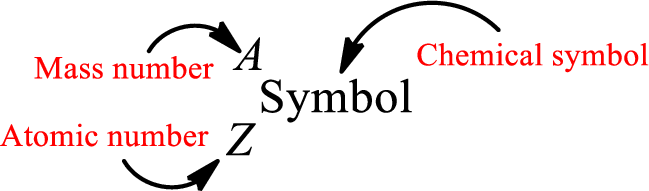
An element is a pure substance that cannot be broken by ordinary chemical reactions into simpler substances. All the atoms in an element will have the same atomic number. The electrons only take part in the chemical reaction while the nucleus does not. Hence, the atomic number (number or protons) does not change and it characterizes an atom.
(b)
Explanation of Solution
Given atom is
Atomic number is given as 78 and mass number is given as 195.
Therefore, the number of protons is 78, number of electrons is 78, and number of neutrons is 117.
The number of protons, electrons and neutrons present in the atom is given.
(c)
Interpretation:
The number of protons, neutrons and electrons present in the atom of
Concept Introduction:
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
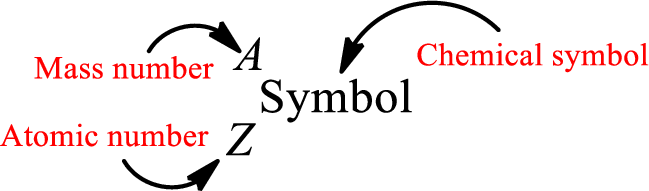
An element is a pure substance that cannot be broken by ordinary chemical reactions into simpler substances. All the atoms in an element will have the same atomic number. The electrons only take part in the chemical reaction while the nucleus does not. Hence, the atomic number (number or protons) does not change and it characterizes an atom.
(c)
Explanation of Solution
Given atom is
Atomic number is given as 31 and mass number is given as 72.
Therefore, the number of protons is 31, number of electrons is 31, and number of neutrons is 41.
The number of protons, electrons and neutrons present in the atom is given.
(d)
Interpretation:
The number of protons, neutrons and electrons present in the atom of
Concept Introduction:
Atomic number for each and every element is a unique one. This is the total number of protons that is present in an atom. As the atom is electrically neutral, it can also be said that the total number of electrons is the atomic number. Atomic number is represented by the symbol Z.
Mass number is the sum of the number of protons and neutrons inside the nucleus of an atom. This gives the number of subatomic particle present inside the nucleus. Mass number is represented by the symbol A.
From atomic number and mass number, the number of each sub atomic particle can be found.
Complete chemical symbol notation can be given as.
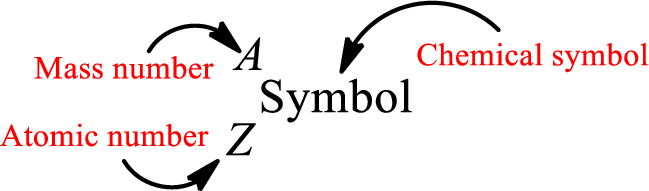
An element is a pure substance that cannot be broken by ordinary chemical reactions into simpler substances. All the atoms in an element will have the same atomic number. The electrons only take part in the chemical reaction while the nucleus does not. Hence, the atomic number (number or protons) does not change and it characterizes an atom.
(d)
Explanation of Solution
Given atom is
Atomic number is given as 12 and mass number is given as 26.
Therefore, the number of protons is 12, number of electrons is 12, and number of neutrons is 14.
The number of protons, electrons and neutrons present in the atom is given.
Want to see more full solutions like this?
Chapter 3 Solutions
General, Organic, and Biological Chemistry
- Which particles in an atom are most responsible for the chemical properties of the atom? Where are these particles located in the atom?arrow_forwardHow many protons and neutrons are contained in the nucleus of each of the following atoms? For an atom of the element, how many electrons are present? a.2963Cu b.3580Br c.1224Mgarrow_forwardHow many electrons, protons, and neutrons arecontained in each atom? a. 55132Cs c. 69163Tm b. 2759Cod. 3070Znarrow_forward
- The number of protons in an atom determines the identity of the atom. What does the number and arrangement of the electrons in an atom determine? What does the number of neutrons in an atom determine?arrow_forwardExplain the operation of a cathode-ray tube. Describe the deflection of cathode rays by electrically charged plates placed within the cathode-ray tube. What does this imply about cathode rays?arrow_forwardComplete the following table. Atmos Number of Protons Number of Neutrons H24e N1020e T2248i O76190s C2750oarrow_forward
- 2.10 Which isotope in each pair contains more neutrons? (A) 35Cl or 33S, (b) 19F or 19Ne, (c) 63Cu or 65Zn, (d) 126I or 127Tearrow_forwardHow many protons and neutrons are contained in the nucleus of each of the following atoms? In an atom of each element, how many electrons are present? a.2241Ti d.3686Kr b.3064Zn e.3375As c.3276Ge f.1941Karrow_forwardHow many protons and electrons are contained in an atom of element 44?arrow_forward
- Fill in the blanks in the table (one column per element).arrow_forwardThe element copper (Cu) has an atomic mass of 63.55 amu and has two naturally occurring isotopic forms. Based on this information, indicate whether each of the following statements is true or false? a. All Cu atoms have a mass of 63.55 amu. b. Some Cu atoms have a mass of 63.55 amu. c. Some Cu atoms have a mass less than 63.55 amu. d. Some Cu atoms have a mass greater than 63.55 amu.arrow_forwardDetermine the number of electrons and protons contained in an atom of the following elements: a. sulfur b. As c. element number 24arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





