
Chemistry In Focus
7th Edition
ISBN: 9781337399692
Author: Tro, Nivaldo J.
Publisher: Cengage Learning,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 65E
Here are three fictitious elements and a molecular view of the atoms that compose them. The molar mass of the middle element, (b), is 25 grams per dozen (g/doz). (The atoms of these fictitious elements are much larger than ordinary atoms.)

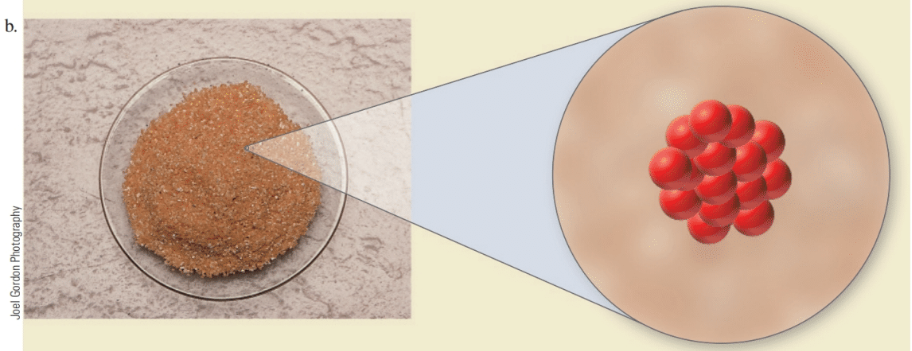

Based on the size of the atoms, do you expect the atomic masses of elements (a) and (c) to be greater than or less than (b)? How many atoms are present in 175 g of element (b)?
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 3 Solutions
Chemistry In Focus
Ch. 3 - Determining the Number of Protons and Electrons in...Ch. 3 - Prob. 3.2YTCh. 3 - Determining Protons, Neutrons, and Electrons How...Ch. 3 - Calculating Atomic Mass Magnesium has three...Ch. 3 - The Mole Concept A diamond, which is pure carbon,...Ch. 3 - The Mole Concept II Calculate the number of atoms...Ch. 3 - Your friend tells you about an article that he...Ch. 3 - Prob. 2SCCh. 3 - Prob. 3SCCh. 3 - Copper is composed of two naturally occurring...
Ch. 3 - Prob. 5SCCh. 3 - Which pair of elements do you expect to be most...Ch. 3 - Which statement is true of the quantum mechanical...Ch. 3 - Why is it important to understand atoms?Ch. 3 - Prob. 2ECh. 3 - What defines an element? How many naturally...Ch. 3 - Prob. 4ECh. 3 - Prob. 5ECh. 3 - Prob. 6ECh. 3 - Prob. 7ECh. 3 - Prob. 8ECh. 3 - Prob. 9ECh. 3 - Prob. 10ECh. 3 - Explain the quantum mechanical model for the atom....Ch. 3 - Give two examples of each: a. alkali metal b....Ch. 3 - Which elements exist as diatomic molecules?Ch. 3 - Explain the difference and similarity between...Ch. 3 - Prob. 15ECh. 3 - Determine the charge of each of the following: a...Ch. 3 - Determine the number of protons and electrons in...Ch. 3 - Determine the number of protons and electrons in...Ch. 3 - Give the atomic number (Z) and the mass number (A)...Ch. 3 - Prob. 20ECh. 3 - The following isotopes have applications in...Ch. 3 - The following isotopes are important in nuclear...Ch. 3 - 14C is used in carbon dating of artifacts....Ch. 3 - 40K is used to measure the age of Earth. Determine...Ch. 3 - Prob. 25ECh. 3 - Prob. 26ECh. 3 - Give electron configurations according to the Bohr...Ch. 3 - Give electron configurations according to the Bohr...Ch. 3 - Prob. 29ECh. 3 - How many valence electrons are in each element of...Ch. 3 - Prob. 31ECh. 3 - Draw electron configurations for each of the...Ch. 3 - Which two of the following elements would you...Ch. 3 - Group the following elements into three similar...Ch. 3 - We have seen that the reactivity of an element is...Ch. 3 - What is the electron configuration of Mg2+? How...Ch. 3 - Classify each of the following elements as a...Ch. 3 - Classify each of the following as a metal, a...Ch. 3 - Calculate the atomic mass of neon (Ne), which is...Ch. 3 - An element has two naturally occurring isotopes....Ch. 3 - A fictitious element has two naturally occurring...Ch. 3 - Copper has two naturally occurring isotopes. Cu-63...Ch. 3 - How many moles of titanium are present in 124 g of...Ch. 3 - Prob. 44ECh. 3 - How many moles are there in each sample? a. 45 mg...Ch. 3 - How many moles are there in each sample? a. 55.0 g...Ch. 3 - What is the mass of each sample? a.1.8 mol S...Ch. 3 - What is the mass of each sample? a.2.75 mol Fe...Ch. 3 - Determine the number of atoms in each sample....Ch. 3 - Determine the number of atoms in each sample....Ch. 3 - How many Ag atoms are present in a piece of pure...Ch. 3 - How many platinum atoms are in a pure platinum...Ch. 3 - A pure gold necklace has a volume of 1.8cm3. How...Ch. 3 - A titanium bicycle component has a volume of...Ch. 3 - An iron sphere has a radius of 3.4 cm. How many...Ch. 3 - Calculate the number of atoms in the universe. The...Ch. 3 - The introduction to this chapter states that...Ch. 3 - Suppose the absolute value of the charge of the...Ch. 3 - When we refer to doughnuts or cookies, we often...Ch. 3 - Prob. 60ECh. 3 - Why does Avogadros number have such an odd value?...Ch. 3 - Prob. 62ECh. 3 - Prob. 63ECh. 3 - Prob. 64ECh. 3 - Here are three fictitious elements and a molecular...Ch. 3 - Prob. 66ECh. 3 - Gather any two of the following items, measure...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Though the common isotope of aluminum has a mass number of 27, isotopes of aluminum have been isolated (or prepared in nuclear reactors) with mass numbers of 24, 25, 26, 28, 29, and 30. How many neutrons are present in each of these isotopes? Why are they all considered aluminum atoms, even though they differ greatly in mass? Write the atomic symbol for each isotope.arrow_forwardA single molecule has a mass of 7.31 1023 g. Provide an example of a real molecule that can have this mass. Assume the elements that make up the molecule are made of light isotopes where the number of protons equals the number of neutrons in the nucleus of each element.arrow_forward2.87 What is the heaviest element to have an atomic weight that is roughly twice its atomic number? What does this suggest must he true about the nuclei of atoms with higher atomic numbers?arrow_forward
- What is the name of the element in Group 4A and Period 5?arrow_forwardWhile traveling to a distant universe, you discover the hypothetical element X. You obtain a representative sample of the element and discover that it is made up of two isotopes, X-23 and X-25. To help your science team calculate the atomic weight of the substance, you send the following drawing of your sample with your report. In the report, you also inform the science team that the brown atoms are X-23, which have an isotopic mass of 23.02 amu, and the green atoms are X-25, which have an isotopic mass of 25.147 amu. What is the atomic weight of element X?arrow_forward2.90 Naturally occurring europium has an average atomic weight of 151.964 amu. If the only isotopes of europium present are 151Eu and 153Eu, describe how you would determine the relative abundance of the two isotopes. Include in your description any information that would need to be looked up.arrow_forward
- What are the different kinds of particles in the atoms nucleus? Compare their properties with each other and with those of an electron.arrow_forwardThe element lithium (Li) has an atomic mass of 6.94 amu and has two naturally occurring istopic forms. Based on this information, indicate whether each of the following statements is true or false? a. All Li atoms have a mass of 6.94 amu. b. Some Li atoms have a mass of 6.94 amu. c. Some Li atoms have a mass of less than 6.94 amu. d. Some Li atoms have a mass of greater than 6.94 amu.arrow_forward2-27 If each atom in Problem 2-26 acquired two more neutrons, what element would each then be?arrow_forward
- The following table presents the abundances and masses of the isotopes of zinc. What is the atomic weight of zinc?arrow_forwardGive the atomic number (Z) and the mass number (A) for each of the following: a. a carbon atom with 8 neutrons b. an aluminum atom with 14 neutrons c. an argon atom with 20 neutrons d. a copper atom with 36 neutronsarrow_forwardCalculate the atomic mass of each of the following elements using the given data for the percentage abundance and mass of each isotope. a. Lithium: 7.42% 6Li (6.01 amu) and 92.58% 7Li (7.02 amu) b. Magnesium: 78.99% 24Mg (23.99 amu), 10.00% 25Mg (24.99 amu), and 11.01% 26Mg (25.98 amu)arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you


 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning



Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning


General, Organic, and Biological Chemistry
Chemistry
ISBN:9781285853918
Author:H. Stephen Stoker
Publisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning
NEET Chemistry | Group 14 Carbon Family | Theory & Problem Solving | In English | Misostudy; Author: Misostudy;https://www.youtube.com/watch?v=enOGIrcHh54;License: Standard YouTube License, CC-BY