
Chemistry & Chemical Reactivity
9th Edition
ISBN: 9781133949640
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3, Problem 91SCQ
A common method for analyzing for the nickel content of a sample is to use a precipitation reaction. Adding the organic compound dimethylgly-oxime to a solution containing Ni2+ ions precipitates a red solid.
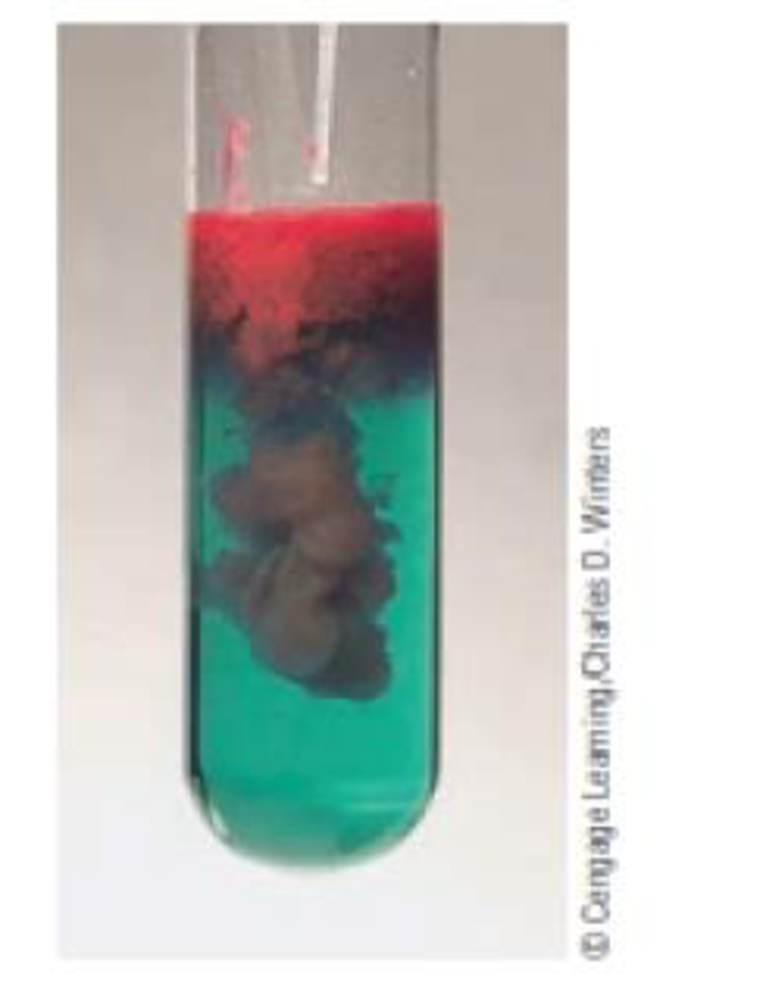
Derive the empirical formula for the red solid based on the following composition: Ni, 20.315%; C, 33.258%; H, 4.884%; 0, 22.151%; and N, 19.392%.
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 3 Solutions
Chemistry & Chemical Reactivity
Ch. 3.1 - The reaction of aluminum with bromine is shown...Ch. 3.1 - If you were to use 8000 atoms of Al, how many...Ch. 3.2 - (a) Butane gas, C4H10, can burn completely in air...Ch. 3.2 - The (unbalanced) equation describing the oxidation...Ch. 3.3 - Prob. 1RCCh. 3.4 - Predict whether each of the following ionic...Ch. 3.4 - Prob. 1RCCh. 3.4 - Prob. 2RCCh. 3.4 - Prob. 3RCCh. 3.5 - In each of the following cases, does a...
Ch. 3.5 - In each of the following cases, aqueous solutions...Ch. 3.5 - Prob. 1RCCh. 3.5 - What is the net ionic equation for the reaction of...Ch. 3.6 - Prob. 1CYUCh. 3.6 - Prob. 2CYUCh. 3.6 - Prob. 1RCCh. 3.6 - 2. The hydrogen phosphate ion is amphiprotic....Ch. 3.6 - What is the net ionic equation for the reaction of...Ch. 3.6 - Prob. 4RCCh. 3.7 - Prob. 1CYUCh. 3.7 - Prob. 1RCCh. 3.8 - Assign an oxidation number to the underlined atom...Ch. 3.8 - Prob. 2CYUCh. 3.8 - 1. What is the oxidation number of Mn in...Ch. 3.8 - Prob. 2RCCh. 3.8 - 3. In which of the manganese compounds below does...Ch. 3.9 - Prob. 1CYUCh. 3.9 - 1. Sometimes a reaction can fall in more than one...Ch. 3.9 - Prob. 1QCh. 3.9 - Prob. 2QCh. 3.9 - Prob. 3QCh. 3.9 - Prob. 4QCh. 3.9 - Prob. 5QCh. 3 - Write balanced chemical equations for the...Ch. 3 - Write balanced chemical equations for the...Ch. 3 - Prob. 3PSCh. 3 - Prob. 4PSCh. 3 - Prob. 5PSCh. 3 - Balance the following equations, and name each...Ch. 3 - Equal amounts of two acidsHCl and HCO2H (formic...Ch. 3 - Prob. 8PSCh. 3 - What is an electrolyte? How can you differentiate...Ch. 3 - Name and give the formulas of two acids that are...Ch. 3 - Which compound or compounds in each of the...Ch. 3 - Which compound or compounds in each of the...Ch. 3 - The following compounds are water-soluble. What...Ch. 3 - The following compounds are water-soluble. What...Ch. 3 - Decide whether each of the following is...Ch. 3 - Decide whether each of the following is...Ch. 3 - Balance the equation for the following...Ch. 3 - Balance the equation for the following...Ch. 3 - Predict the products of each precipitation...Ch. 3 - Prob. 20PSCh. 3 - Write a balanced equation for the ionization of...Ch. 3 - Write a balanced equation for the ionization of...Ch. 3 - Prob. 23PSCh. 3 - Phosphoric add can supply one, two, or three H3O+...Ch. 3 - Prob. 25PSCh. 3 - Prob. 26PSCh. 3 - Prob. 27PSCh. 3 - Prob. 28PSCh. 3 - Prob. 29PSCh. 3 - Prob. 30PSCh. 3 - Write an equation that describes the equilibrium...Ch. 3 - Write an equation that describes the equilibrium...Ch. 3 - Prob. 33PSCh. 3 - Write two chemical equations, one in which H2PO4...Ch. 3 - Balance the following equations, and then write...Ch. 3 - Balance the following equations, and then write...Ch. 3 - Prob. 37PSCh. 3 - Balance each of the following equations, and then...Ch. 3 - Write balanced net ionic equations for the...Ch. 3 - Write balanced net ionic equations for the...Ch. 3 - Siderite is a mineral consisting largely of...Ch. 3 - The mineral rhodothrosite is manganese()...Ch. 3 - Prob. 43PSCh. 3 - Prob. 44PSCh. 3 - Determine the oxidation number of each element in...Ch. 3 - Determine the oxidation number of each element in...Ch. 3 - Prob. 47PSCh. 3 - Which two of the following reactions are...Ch. 3 - In the following reactions, decide which reactant...Ch. 3 - In the following reactions, decide which reactant...Ch. 3 - Balance the following equations, and then classify...Ch. 3 - Prob. 52PSCh. 3 - Classify each of the following reactions as a...Ch. 3 - Prob. 54PSCh. 3 - Balance each of the following equations, and...Ch. 3 - Complete and balance the equations below, and...Ch. 3 - Prob. 57PSCh. 3 - Prob. 58PSCh. 3 - Balance the following equations: (a) for the...Ch. 3 - Balance the following equations: (a) for the...Ch. 3 - Prob. 61GQCh. 3 - Give the formula for each of the following...Ch. 3 - Prob. 63GQCh. 3 - Name two anions that combine with Al3+ ion to...Ch. 3 - Write the net ionic equation and identify the...Ch. 3 - Identify and name the water-insoluble product in...Ch. 3 - Bromine is obtained from sea water by the...Ch. 3 - Identify each of the blowing substances as a...Ch. 3 - The mineral dolomite contains magnesium...Ch. 3 - Aqueous solutions of ammonium sulfide, (NH4)2S,...Ch. 3 - Prob. 71GQCh. 3 - Prob. 72GQCh. 3 - Balance equations for these reactions that occur...Ch. 3 - Prob. 74GQCh. 3 - You are given mixtures containing the following...Ch. 3 - Identify, from each list below, the compound or...Ch. 3 - Prob. 77GQCh. 3 - Prob. 78GQCh. 3 - Gas evolution was observed when a solution of Na2S...Ch. 3 - Prob. 81ILCh. 3 - Prob. 82ILCh. 3 - Prob. 83ILCh. 3 - A Suggest a laboratory method for preparing barium...Ch. 3 - The Toliens test for the presence of reducing...Ch. 3 - There are many ionic compounds that dissolve in...Ch. 3 - Most naturally occurring acids are weak acids....Ch. 3 - You want to prepare barium chloride, BaC12, using...Ch. 3 - Prob. 89SCQCh. 3 - A Describe how to prepare zinc chloride by (a) an...Ch. 3 - A common method for analyzing for the nickel...Ch. 3 - The presence of arsenic in a sample that may also...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Iron oxide ores, commonly a mixture of FeO and Fe2O3, are given the general formula Fe3O4. They yield elemental iron when heated to a very high temperature with either carbon monoxide or elemental hydrogen. Balance the following equations for these processes: Fe3O4(s)+H2(g)Fe(s)+H2O(g)Fe3O4(s)+CO(g)Fe(s)+CO2(g)arrow_forwardThe balanced equation for the reduction of iron ore to the metal using CO is Fe2O3(s) + 3 CO(g) 2 Fe(s) + 3 CO2(g) (a) What is the maximum mass of iron, in grams, that can be obtained from 454 g (1.00 lb) of iron(III) oxide? (b) What mass of CO is required to react with 454 g cot Fe2O3?arrow_forwardNitric acid is produced commercially by the Ostwald process, represented by the following equations: 4NH3(g)+5O24NO(g)+6H2O(g)2NO(g)+O2(g)2NO2(g)3NO2(g)+H2O(l)2HNO3(aq)+NO(g) What mass of NH3 must be used to produce 1.0 106 kg HNO3 by the Ostwald process? Assume 100% yield in each reaction, and assume that the NO produced in the third step is not recycled.arrow_forward
- Write a balanced equation for (a) the combustion (reaction with oxygen gas) of glucose, C6H12O6, to give carbon dioxide and water. (b) the reaction between xenon tetrafluoride gas and water to give xenon, oxygen, and hydrogen fluoride gases. (c) the reaction between aluminum and iron(III) oxide to give aluminum oxide and iron. (d) the formation of ammonia gas from its elements. (e) the reaction between sodium chloride, sulfur dioxide gas, steam, and oxygen to give sodium sulfate and hydrogen chloride gas.arrow_forwardBacterial digestion is an economical method of sewage treatment. The reaction is an intermediate step in the conversion of the nitrogen in organic compounds into nitrate ions. What mass of bacterial tissue is produced in a treatment plant for every 1.0 104 kg of wastewater containing 3.0% NH4+ ions by mass? Assume that 95% of the ammonium ions are consumed by the bacteria.arrow_forwardSilicon is produced for the chemical and electronics industries by the following reactions. Give the balanced equation for each reaction. a. SiO2(s)+C(s)arefurnaceElectricSi(s)+CO(g) b. Liquid silicon tetrachloride is reacted with very pure solid magnesium, producing solid silicon and solid magnesium chloride. c. Na2SiF6(s) + Na(s) Si(s) + NaF(s)arrow_forward
- The present average concentration (mass percent) of magnesium ions in seawater is 0.13%. A chemistry textbook estimates that if 1.00 × 108 tons Mg were taken out of the sea each year, it would take one million years for the Mg concentration to drop to 0.12%. Do sufficient calculations to either verify or refute this statement. Assume that Earth is a sphere with a diameter of 8000 mi, 67% of which is covered by oceans to a depth of 1 mi, and that no Mg is washed back into the oceans at any time.arrow_forwardYou take 1.00 g of an aspirin tablet (a compound consisting solely of carbon, hydrogen, and oxygen), burn it in air, and collect 2.20 g CO2 and 0.400 g H2O. You know that the molar mass of aspirin is between 170 and 190 g/mol. Reacting 1 mole of salicylic acid with I mole of acetic anhydride (C4H6O3) gives you 1 mole of aspirin and 1 mole of acetic acid (C2H4O2). Use this information to determine the molecular formula of salicylic acid.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
Step by Step Stoichiometry Practice Problems | How to Pass ChemistryMole Conversions Made Easy: How to Convert Between Grams and Moles; Author: Ketzbook;https://www.youtube.com/watch?v=b2raanVWU6c;License: Standard YouTube License, CC-BY