
a) Syndiotactic polyacrylonitrile
Interpretation:
Draw the three-dimensional structure of the polymeric products given in the question.
Concept introduction:
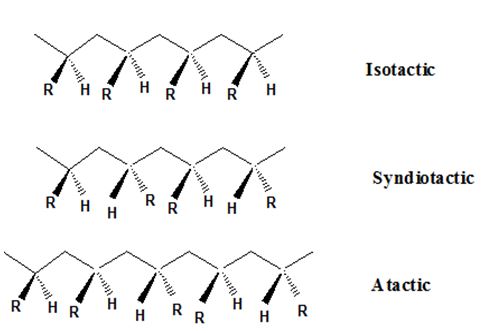
Isotactic, syndiotactic, and atactic are the stereochemical forms. The
Syndiotactic are the macromolecules in which the (-R) groups are arranged in an alternate manner along the long chain of the polymer. Gutta percha is also an example for Syndiotactic polymer.
In atactic form, the substituents are placed in a random manner along the long chain.
The important point to note here is that the polymer obtained from the chain growth
b) Atactic poly(methyl methacrylate)
Interpretation:
Draw the three-dimensional structure of the polymeric products given in the question.
Concept introduction:
Isotactic, syndiotactic, and atactic are the stereochemical forms. The polymer having the (-R) group on the same side of the macromolecule backbone (zig-zag bone) is known as isotactic polymer. An isotactic macromolecule consists of 100% meso diads.
Syndiotactic are the macromolecules in which the (-R) groups are arranged in an alternate manner along the long chain of the polymer. Gutta percha is also an example for Syndiotactic polymer.
In atactic form, the substituents are placed in a random manner along the long chain.
The important point to note here is that the polymer obtained from the chain growth polymerization mechanism must have chiral carbons to possess these structures. If the polymer formed does not have chiral carbon then it cannot occur in isotactic, syndiotactic and atactic forms.

c) Isotactic poly(vinyl chloride)
Interpretation:
Draw the three-dimensional structure of the polymeric products given in the question.
Concept introduction:
Isotactic, syndiotactic, and atactic are the stereochemical forms. The polymer having the (-R) group on the same side of the macromolecule backbone (zig-zag bone) is known as isotactic polymer. An isotactic macromolecule consists of 100% meso diads.
Syndiotactic are the macromolecules in which the (-R) groups are arranged in an alternate manner along the long chain of the polymer. Gutta percha is also an example for Syndiotactic polymer.
In atactic form, the substituents are placed in a random manner along the long chain.
The important point to note here is that the polymer obtained from the chain growth polymerization mechanism must have chiral carbons to possess these structures. If the polymer formed does not have chiral carbon then it cannot occur in isotactic, syndiotactic and atactic forms.

Want to see the full answer?
Check out a sample textbook solution
Chapter 31 Solutions
Organic Chemistry
- Proteins are naturally occurring polymers formed by condensationreactions of amino acids, which have the generalstructureIn this structure, ¬R represents ¬H, ¬CH3, or anothergroup of atoms; there are 20 different natural amino acids,and each has one of 20 different R groups. (a) Draw the generalstructure of a protein formed by condensation polymerizationof the generic amino acid shown here. (b) When only afew amino acids react to make a chain, the product is called a“peptide” rather than a protein; only when there are 50 aminoacids or more in the chain would the molecule be called a protein.For three amino acids (distinguished by having three differentR groups, R1, R2, and R3), draw the peptide that resultsfrom their condensation reactions. (c) The order in which theR groups exist in a peptide or protein has a huge influence onits biological activity. To distinguish different peptides andproteins, chemists call the first amino acid the one at the “Nterminus” and the last one the one at…arrow_forwardWhich of the following provides an absolute measure of the molecular weight of polymers:(a) viscometry, (b) cryometry, (c) osmometry, (d) light-scattering photometry, (e) GCP?arrow_forwardProteins are naturally occurring polymers formed by condensationreactions of amino acids, which have the generalstructure In this structure, — R represents — H, — CH3, or anothergroup of atoms; there are 20 different natural amino acids,and each has one of 20 different R groups. (a) Draw the generalstructure of a protein formed by condensation polymerizationof the generic amino acid shown here. (b) When only afew amino acids react to make a chain, the product is called a“peptide” rather than a protein; only when there are 50 aminoacids or more in the chain would the molecule be called a protein.For three amino acids (distinguished by having three differentR groups, R1, R2, and R3), draw the peptide that resultsfrom their condensation reactions. (c) The order in which theR groups exist in a peptide or protein has a huge influence onits biological activity. To distinguish different peptides andproteins, chemists call the first amino acid the one at the “Nterminus” and the last one the…arrow_forward
- Organic chemistry: brief introduction of interfacial polymerization by nylonarrow_forward(a)Is this a condensation or addition polymer? Explain how you know. (b) Provide the IUPAC name of the monomer that does not contain the benzene group.arrow_forwardOutline some methods for obtaining ultra-high orientation in polymers (e.g. Kevlar).arrow_forward
- When Cationic polymerization occurs ? & how it occurs ?arrow_forwardIn the polymerization of polyethelene, the monomer is the ______________.arrow_forward5. 1) Discuss how polymerization changes the properties of any ensemble of amino acids so that a protein polymer functions essentially differently than an aggregate of amino acids. 2) Why does a protein need to be a polymer in order to properly function??arrow_forward
- 1. Explain the following observations: (i) Kevlar has an extremely high tensile strength and melting temperature. (ii) At room temperature, poly(methylmethacrylate) is transparent, while polyethylene istranslucent. At temperatures above 135°C, both polymers are transparent. (iii) Polyoxymethylene and polyethylene exhibit similar mechanical behaviour. However, the glass transition temperature of polyoxymethylene is about 50°C higher than that of polyethylene.arrow_forwardAn alternating copolymer is known to have a number-average molecular weight of 100,000 g/mol and a degree of polymerization of 2210. If one of the repeat units is ethylene, which of styrene, propylene, tetrafluoroethylene, and vinyl chloride is the other repeat unit? Why?arrow_forwardSEE THE ATTACHED IAMGEarrow_forward

 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning




