
Concept explainers
(a)
Interpretation:
Whether the given set are considered as valid pairs of contributing structures or not.
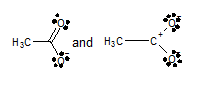
Concept Introduction:
Resonance is the method which explains electron’s delocalization in the molecules. It contrasts and compares more than two Lewis strictures which can signify the specific molecule. When a single molecule couldn’t explain the proper bonding between the two atoms so for explaining the proper bonding between two atoms resonance structure is used in place of Lewis structure.
(b)
Interpretation:
Whether the given set are considered as valid pairs of contributing structures or not.
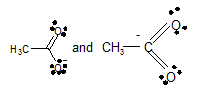
Concept Introduction:
In
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Introduction to General, Organic and Biochemistry
- Problem 3-6 Give each binary compound a systematic name and a common name. (a) FeO (b) Fe2O3arrow_forwardProblem 3-4 Name these binary ionic compounds: (a) MgO ( b) BaI2 (c) KClarrow_forwardConvert the following representation of ethane, C2H6, into a conventional drawing that uses solid, wedged, and dashed lines to indicate tetrahedral geometry around each carbon (gray = C, ivory = H).arrow_forward
- Problem 3-7 Name these ionic compounds, each of which contains a polyatomic ion: (a) K2HPO4 (b) Al2(SO4)3 (c) FeCO3arrow_forwardProblem 3-2 Judging from their relative positions in the Periodic Table, which element in each pair has the larger electronegativity? (a) Lithium or potassium (b) Nitrogen or phosphorus (c) Carbon or siliconarrow_forwardProblem 3-15 Predict all bond angles for these molecules: (a) CH3OH (b) CH2Cl2 (c) H2CO3 (carbonic acid)arrow_forward
- Draw the indicated number of resonance forms for each of the following species: (a) The methyl phosphate anion, CH3OPO32- (3) (b) The nitrate anion, NO3- (3) (c) The allyl cation, H2C=CH—CH2+ (2) (d) The benzoate anion (4)arrow_forwardProblem 3-1 Show how the following chemical changes obey the octet rule: (a) A magnesium atom forms a magnesium ion, Mg2+. (b) A sulfur atom forms a sulfide ion, S2-.arrow_forwardProblem 3-16 Which of these molecules are polar? Show the direction of the molecular dipole by using an arrow with a crossed tail. (a) H2S (b) HCN (C) C2H6arrow_forward
 Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning


