
Chemistry & Chemical Reactivity
10th Edition
ISBN: 9781337399074
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 4, Problem 130IL
Sulfuric acid is listed in a catalog with a concentration of 95-98%. A bottle of the acid in the stockroom states that 1.00 L has a mass of 1.84 kg. To determine the concentration of sulfuric acid in the stockroom bottle, a student dilutes 5.00 mL to 500. mL She then takes four 10.00-mL samples and titrates each with standardized sodium hydroxide (c = 0.1760 M).
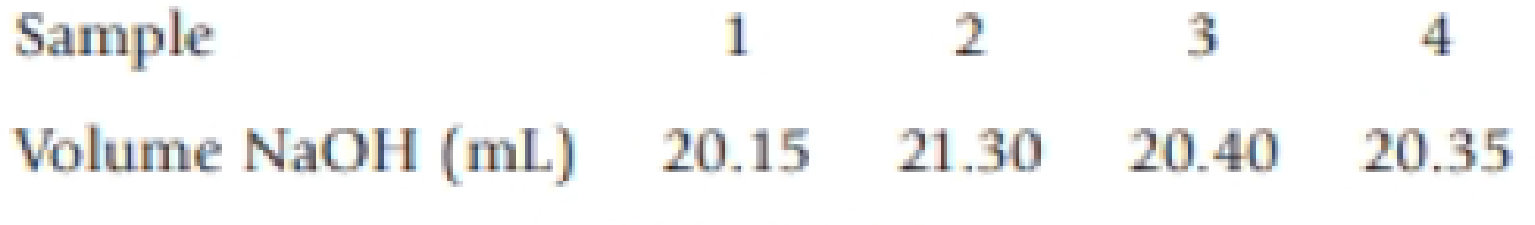
- (a) What is the average concentration of the diluted sulfuric acid sample?
- (b) What is the mass percent of H2SO4 in the original bottle of the acid?
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 4 Solutions
Chemistry & Chemical Reactivity
Ch. 4.1 - What mass of oxygen, O2, is required to completely...Ch. 4.2 - The thermite reaction produces iron metal and...Ch. 4.4 - One method for determining the purity of a sample...Ch. 4.4 - A 0523-g sample of the unknown compound CxHy was...Ch. 4.4 - Prob. 4.5CYUCh. 4.5 - Sodium bicarbonate, NaHCO3, is used in baking...Ch. 4.5 - An experiment calls for you to use 250. mL of 1.00...Ch. 4.6 - (a) What is the pH of a solution of HCI in which...Ch. 4.7 - Prob. 4.9CYUCh. 4.8 - A 25.0-mL sample of vinegar (which contains the...
Ch. 4.8 - Hydrochloric acid. HCl, with a concentration of...Ch. 4.8 - An unknown monoprotic acid reacts with NaOH...Ch. 4.8 - Vitamin C, ascorbic acid (C6HgO6)(molar mass 176.1...Ch. 4.9 - Prob. 4.14CYUCh. 4.9 - Prob. 1.1ACPCh. 4.9 - Excess KI is added to a 100.0-mL sample of a soft...Ch. 4.9 - Prob. 3.1ACPCh. 4.9 - Identify the factor labeled 4 in the strategy...Ch. 4.9 - Identify the factor labeled 3 in this strategy...Ch. 4.9 - Prob. 4.3ACPCh. 4.9 - Prob. 4.4ACPCh. 4 - The reaction of iron(III) oxide with aluminum to...Ch. 4 - What mass of HCI, in grams, is required to react...Ch. 4 - Like many metals, aluminum reacts with a halogen...Ch. 4 - The balanced equation for the reduction of iron...Ch. 4 - Methane, CH4, burns in oxygen. (a) What are the...Ch. 4 - The formation of water-Insoluble silver chloride...Ch. 4 - The metals industry was a major source of air...Ch. 4 - Prob. 8PSCh. 4 - Chromium metal reacts with oxygen to give...Ch. 4 - Ethane, C2H6, burns in oxygen. (a) What are the...Ch. 4 - Prob. 11PSCh. 4 - Ammonia gas can be prepared by the reaction of a...Ch. 4 - The compound SF6 is made by burning sulfur in an...Ch. 4 - Disulfur dichloride, S2Cl2, is used to vulcanize...Ch. 4 - The reaction of methane and water is one way to...Ch. 4 - Aluminum chloride AlCl3, is made by treating scrap...Ch. 4 - In the thermite reaction, iron(III) oxide is...Ch. 4 - Aspirin, C6H4 (OCOCH3) CO3H, is produced by the...Ch. 4 - In Example 4.2, you found that a particular...Ch. 4 - Ammonia gas can be prepared by the following...Ch. 4 - The deep blue compound Cu(NH3)4S04 is made by the...Ch. 4 - Black smokers are found in the depths of the...Ch. 4 - The reaction of methane and water is one way to...Ch. 4 - Methanol, CH3OH, can be prepared from carbon...Ch. 4 - A mixture of CuSO4 and CuSO4.5 H2O has a mass of...Ch. 4 - A 2.634-g sample containing impure CuCl2 2 H2O was...Ch. 4 - Prob. 27PSCh. 4 - Prob. 28PSCh. 4 - Nickel(II) sulfide, NiS, occurs naturally as the...Ch. 4 - The aluminum in a 0.764-g sample of an unknown...Ch. 4 - Prob. 31PSCh. 4 - Mesitylene is a liquid hydrocarbon Burning 0.115 g...Ch. 4 - Naphthalene is a hydrocarbon that once was used in...Ch. 4 - Azulene is a beautiful blue hydrocarbon. If 0.106...Ch. 4 - An unknown compound has the formula CxHyOz. You...Ch. 4 - An unknown compound has the formula CxHyOz. You...Ch. 4 - Nickel forms a compound with carbon monoxide,...Ch. 4 - To find the formula of a compound composed of iron...Ch. 4 - If 6.73 g of Na2CO3 is dissolved in enough water...Ch. 4 - Some potassium dichromate (K2Cr2O7), 2.335 g, is...Ch. 4 - What is the mass of solute, in grams, in 250, mL...Ch. 4 - Prob. 42PSCh. 4 - What volume of 0123 M NaOH, in milliliters,...Ch. 4 - What volume of 2.06 M KMnO4, in liters, contains...Ch. 4 - Identify the ions that exist in each aqueous...Ch. 4 - Identify the ions that exist in each aqueous...Ch. 4 - An experiment in your laboratory requires 500. mL...Ch. 4 - What mass of oxalic acid, H2C2O4, is required to...Ch. 4 - If you dilute 25.0 mL of 1.50 M hydrochloric acid...Ch. 4 - If 4.00 mL of 0.0250 M CuSO4 is diluted to 10.0 mL...Ch. 4 - Which of the following methods would you use to...Ch. 4 - Which of the following methods would you use to...Ch. 4 - You have 250. mL of 0.136 M HCl. Using a...Ch. 4 - Prob. 54PSCh. 4 - A table wine has a pH of 3.40. What is the...Ch. 4 - A saturated solution of milk of magnesia, Mg(OH)2,...Ch. 4 - Prob. 57PSCh. 4 - Prob. 58PSCh. 4 - Prob. 59PSCh. 4 - Prob. 60PSCh. 4 - Prob. 61PSCh. 4 - What mass of Na2CO3, in grams, is required for...Ch. 4 - When an electric current is passed through an...Ch. 4 - Hydrazine, N2H4, a base like ammonia, can react...Ch. 4 - In the photographic developing process, silver...Ch. 4 - You can dissolve an aluminum soft drink can in an...Ch. 4 - What volume of 0.750 M Pb(NO3)2, in milliliters,...Ch. 4 - What volume of 0.125 M oxalic acid, H2C2O4, is...Ch. 4 - What volume of 0.812 M HCI, in milliliters, is...Ch. 4 - What volume of 0.955 M HCl, in milliliters, is...Ch. 4 - If 38.55 mL of HCI is required to titrate 2.150 g...Ch. 4 - Potassium hydrogen phthalate, KHCgH4O4, is used to...Ch. 4 - You have 0.954 g of an unknown acid, H2A, which...Ch. 4 - An unknown solid acid is either citric acid or...Ch. 4 - To analyze an iron-containing compound, you...Ch. 4 - Vitamin C has the formula C6H8O6. Besides being an...Ch. 4 - Prob. 77PSCh. 4 - Suppose 16.04 g of benzene, C6H6, is burned in...Ch. 4 - The metabolic disorder diabetes causes a buildup...Ch. 4 - Your body deals with excess nitrogen by excreting...Ch. 4 - The reaction of iron metal and chlorine gas to...Ch. 4 - Prob. 83GQCh. 4 - The reaction of 750. g each of NH3 and O2 was...Ch. 4 - Sodium azide, an explosive chemical used in...Ch. 4 - Prob. 86GQCh. 4 - Prob. 87GQCh. 4 - Prob. 88GQCh. 4 - Prob. 89GQCh. 4 - A Menthol, from oil of mint, has a characteristic...Ch. 4 - Benzoquinone, a chemical used in the dye industry...Ch. 4 - Aqueous solutions of iron(II) chloride and sodium...Ch. 4 - Sulfuric acid can be prepared starting with the...Ch. 4 - Prob. 94GQCh. 4 - An unknown metal reacts with oxygen to give the...Ch. 4 - Titanium(IV) oxide, TiO2, is heated in hydrogen...Ch. 4 - Potassium perchlorate is prepared by the following...Ch. 4 - A Commercial sodium "hydrosulfite" is 90.1%...Ch. 4 - What mass of lime, CaO, can be obtained by heating...Ch. 4 - The elements silver, molybdenum, and sulfur...Ch. 4 - A mixture of butene, C4Hg, and butane, is burned...Ch. 4 - Cloth can be waterproofed by coating it with a...Ch. 4 - Copper metal can be prepared by roasting copper...Ch. 4 - Prob. 104GQCh. 4 - Sodium bicarbonate and acetic acid react according...Ch. 4 - A noncarbonated soft drink contains an unknown...Ch. 4 - Sodium thiosulfate, Na2S2O3, is used as a fixer in...Ch. 4 - You have a mixture of oxalic acid, H2C2O4, and...Ch. 4 - (a) What is the pH of a 0.105 M HCl solution? (b)...Ch. 4 - A solution of hydrochloric acid has a volume of...Ch. 4 - One half liter (500. mL) of 2.50 M HCl is mixed...Ch. 4 - A solution of hydrochloric acid has a volume of...Ch. 4 - Prob. 113GQCh. 4 - Prob. 115GQCh. 4 - Prob. 116GQCh. 4 - Gold can be dissolved from gold-bearing rock by...Ch. 4 - You mix 25.0 mL of 0.234 M FeCl3 with 42.5 mL of...Ch. 4 - Prob. 119GQCh. 4 - ATOM ECONOMY: Ethylene oxide, C2H4O, is an...Ch. 4 - Suppose you dilute 25.0 mL of a 0.110 M solution...Ch. 4 - Prob. 122ILCh. 4 - Oyster beds in the oceans require chloride ions...Ch. 4 - You wish to determine the weight percent of copper...Ch. 4 - Prob. 126ILCh. 4 - Chromium(III) chloride forms many compounds with...Ch. 4 - Thioridazine, C21H26N2S2, is a pharmaceutical...Ch. 4 - A herbicide contains 2,4-D...Ch. 4 - Sulfuric acid is listed in a catalog with a...Ch. 4 - Two beakers sit on a balance; the total mass is...Ch. 4 - A weighed sample of iron (Fe) is added to liquid...Ch. 4 - Let us explore a reaction with a limiting...Ch. 4 - Two students titrate different samples of the same...Ch. 4 - ATOM ECONOMY: Benzene, C6H6, is a common compound,...Ch. 4 - ATOM ECONOMY: Maleic anhydride, C4H2O3, can be...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Arsenic acid, H3AsO4, is a poisonous acid that has been used in the treatment of wood to prevent insect damage. Arsenic acid has three acidic protons. Say you take a 25.00-mL sample of arsenic acid and prepare it for titration with NaOH by adding 25.00 mL of water. The complete neutralization of this solution requires the addition of 53.07 mL of 0.6441 M NaOH solution. Write the balanced chemical reaction for the titration, and calculate the molarity of the arsenic acid sample.arrow_forwardVitamin C is ascorbic acid, HC6H7O6, which can be titrated with a strong base. HC6H7O6(aq) + NaOH(aq) NaC6H7O6(aq) + H2O() A student dissolved a 500.0-mg vitamin C tablet in 200.0 mL water and then titrated it with 0.1250-M NaOH. It required 21.30 mL of the base to reach the equivalence point. Calculate the mass percentage of the tablet that is impurity.arrow_forwardA 1.345-g sample of a compound of barium and oxygen was dissolved in hydrochloric acid to give a solution of barium ion, which was then precipitated with an excess of potassium chromate to give 2.012 g of barium chromate, BaCrO4. What is the formula of the compound?arrow_forward
- Twenty-five mL of a 0.388 M solution of Na2SO4 is mixed with 35.3 mL of 0.229 M Na2SO4. What is the molarity of the resulting solution? Assume that the volumes are additive.arrow_forwardPotatoes can be peeled commercially by soaking them in a 3-M to 6-M solution of sodium hydroxide, then removing the loosened skins by spraying them with water. Does a sodium hydroxide solution have a suitable concentration if titration of 12.00 mL of the solution requires 30.6 mL of 1.65 M HCI to reach the end point?arrow_forwardBone was dissolved in hydrochloric acid, giving 50.0 mL of solution containing calcium chloride, CaCL2. To precipitate the calcium ion from the resulting solution, an excess of potassium oxalate was added. The precipitate of calcium oxalate, CaC2O4, weighed 1.437 g. What was the molarity of CaCl2 in the solution?arrow_forward
- An antacid tablet contains sodium hydrogen carbonate, NaHCO3, and inert ingredients. A 0.465-g sample of powdered tablet was mixed with 53.3 mL of 0.190 M HCl (hydrochloric acid). The mixture was allowed to stand until it reacted. NaHCO3(s)+HCl(aq)NaCl(aq)+H2O(l)+CO2(g) The excess hydrochloric acid was titrated with 54.6 mL of 0.128 M NaOH (sodium hydroxide). HCl(aq)+NaOH(aq)NaCl(aq)+H2O(l) What is the percentage of sodium hydrogen carbonate in the antacid?arrow_forwardA 0.608-g sample of fertilizer contained nitrogen as ammonium sulfate, (NH4)2SO4. It was analyzed for nitrogen by heating with sodium hydroxide. (NH4)2SO4(s)+2NaOH(aq)Na2SO4(aq)+2H2O(l)+2NH3(g) The ammonia was collected in 46.3 mL of 0.213 M HCl (hydrochloric acid), with which it reacted. NH3(g)+HCl(aq)NH4Cl(aq) This solution was titrated for excess hydrochloric acid with 44.3 mL of 0.128 M NaOH. NaOH(aq)+HCl(aq)NaCl(aq)+H2O(l) What is the percentage of nitrogen in the fertilizer?arrow_forwardCitric acid, which can be obtained from lemon juice, has the molecular formula C6H8O7. A 0.250-g sample of citric acid dissolved in 25.0 mL of water requires 37.2 mL of 0.105 M NaOH for complete neutralization. What number of acidic hydrogens per molecule does citric acid have?arrow_forward
- The active ingredients of an antacid tablet contained only magnesium hydroxide and aluminum hydroxide. Complete neutralization of a sample of the active ingredients required 48.5 mL of 0.187 M hydrochloric acid. The chloride salts from this neutralization were obtained by evaporation of the filtrate from the titration; they weighed 0. 4200 g. What was the percentage by mass of magnesium hydroxide in the active ingredients of the antacid tablet?arrow_forwardA soluble iodide was dissolved in water. Then an excess of silver nitrate, AgNO3, was added to precipitate all of the iodide ion as silver iodide, AgI. If 1.545 g of the soluble iodide gave 2.185 g of silver iodide, how many grams of iodine are in the sample of soluble iodide? What is the mass percentage of iodine, I, in the compound?arrow_forwardElemental bromine is the source of bromine compounds. The element is produced from certain brine solutions that occur naturally. These brines are essentially solutions of calcium bromide that, when treated with chlorine gas, yield bromine in a displacement reaction. What are the molecular equation and net ionic equation for the reaction? A solution containing 40.0 g of calcium bromide requires 14.2 g of chlorine to react completely with it, and 22.2 g of calcium chloride is produced in addition to whatever bromine is obtained. How many grams of calcium bromide are required to produce 10.0 pounds of bromine?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning
Acid-Base Titration | Acids, Bases & Alkalis | Chemistry | FuseSchool; Author: FuseSchool - Global Education;https://www.youtube.com/watch?v=yFqx6_Y6c2M;License: Standard YouTube License, CC-BY