
Concept explainers
- a. Draw three constitutional isomers with molecular formula C3H8O.
- b. How many constitutional isomers can you draw for C4H10O?
(a)
Interpretation:
The three constitutional isomers of molecular formula
Concept introduction:
Constitutional isomers are having the same molecular formula and different bond connectivity between the atoms.
The general formula for an alkane is
Equation for finding constitutional isomers for a molecule
Answer to Problem 1P
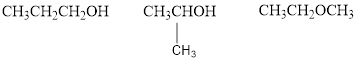
Explanation of Solution
The given molecular formula is
Therefore,
The two possible alcohols are,
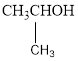
The possible ether is,
(b)
Interpretation:
The numbers of constitutional isomers of molecular formula
Concept introduction:
Constitutional isomers are having the same molecular formula and different bond connectivity between the atoms.
Answer to Problem 1P
The numbers of constitutional isomers of molecular formula
Explanation of Solution
The given molecular formula is
Therefore,
There will be five alcohol isomers (due to four carbons) and two ether isomers.
Hence,
Want to see more full solutions like this?
Chapter 4 Solutions
Essential Organic Chemistry, Global Edition
Additional Science Textbook Solutions
Chemistry
Inorganic Chemistry
Introductory Chemistry (5th Edition) (Standalone Book)
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
- Draw the five constitutional isomers that have molecular formula C5H10 and contain one ring ?arrow_forwardDraw three constitutional isomers having molecular formula C7H14 that contains a five-membered ring and two methyl groups bonded to that ring.arrow_forward(a) Using Newman projections, draw all staggered and eclipsed conformations that result from rotation around the bond highlighted in red in each molecule; (b) draw a graph of energy versus dihedral angle for rotation around this bond.arrow_forward
- Identify the type of configurational isomerism that exist for each double bond. E or Zarrow_forwardAssign the absolute configuration of all molecules. Draw the most stable conformer in Newman projects with respect to bond C2-C3; for both a) and b). Draw Fisher projections for all molecules.arrow_forwardAssign the absolute configuration of all molecules. Draw the most stable conformer in Newman projects with respect to bond C2-C3; for both a) and b). Draw Fisher projections for all molecules. do in detail completearrow_forward
- How many tetrahedral stereogenic centers does PGF2a contain? Draw its enantiomer. How many of its double bonds can exhibit cis-trans isomerism? Considering both its double bonds and its tetrahedral stereogenic centers, how many stereoisomers are possible for PGF2a?arrow_forwardDraw the three constitutional isomers having molecular formula C7H14 that contain a five-membered ring and two methyl groups as substituents. For each constitutional isomer that can have cis and trans isomers, draw the two stereoisomers.arrow_forwardFor each compound drawn below: a.Label each OH, Br, and CH3 group as axial or equatorial. b.Classify each conformation as cis or trans.c.Translate each structure into a representation with a hexagon for the six-membered ring, and wedges and dashed wedges for groups above and below the ring. d. Draw the second possible chair conformation for each compound.arrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

