
(a)
Interpretation:
The given carbon oxygen-containing compound has a carbonyl group or not has to be indicated.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom.
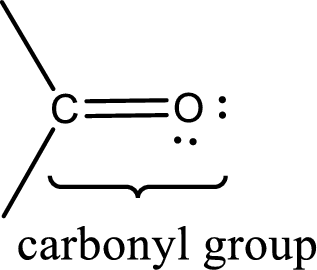
(b)
Interpretation:
The given carbon oxygen-containing compound has a carbonyl group or not has to be indicated.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
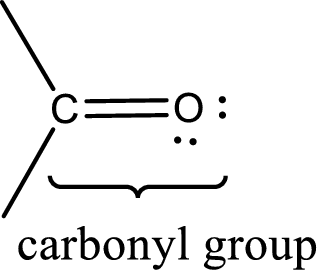
(c)
Interpretation:
The given carbon oxygen-containing compound has a carbonyl group or not has to be indicated.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
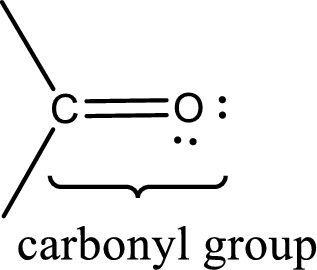
(d)
Interpretation:
The given carbon oxygen-containing compound has a carbonyl group or not has to be indicated.
Concept Introduction:
Carbonyl groups are the one which contain a double bond between carbon and oxygen atom. Aldehydes and ketones possess this carbonyl functional group in it. The structural representation of a carbonyl group can be given as shown below,
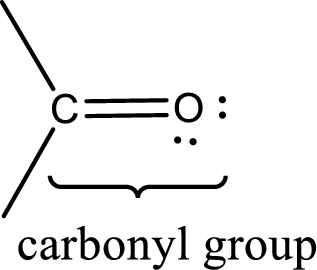
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
Organic And Biological Chemistry
- Draw condensed structural formulas for the following. a. All ethers that are functional group isomers of 2-methyl-1-propanol b. All alcohols that are functional group isomers of 1-ethoxyethanearrow_forwardWhich of the following organic compounds has a functional group of a carboxyl group?A. alkyl halide B. ester C. ketone D. aminearrow_forwardWhat structural characteristic is shared by the aldehydes and the ketones? A) They both are straight chain compounds. B) Aldehydes and ketones both contain a carbonyl carbon. C) Both of these compound classes have as the smallest compound a 5 carbon skeleton. D) Aldehydes and ketones have no shared characteristics.arrow_forward
- What is the functional isomer of butanal? butanoic acid 1-butanol butanone methyl propanoate Other:arrow_forwardWrite structural formulas for the following compounds.(a) ethyl isopropyl ether (b) di-n-butyl etherarrow_forwardName the functional group present in the following molecule: C2H5COCH3 Select one: 1- Alcohol 2- Ether 3- Aldehyde 4- Ketonearrow_forward
- What is the functional group of the following compound A. Ketone B.ester C.amide D.etherarrow_forwardFor embalming purposes, phenol has the properties of preserving, disinfecting, and ___ . cauterizing cut tissues adding a pink glow to the body adding a nice fragrance anticoagulantarrow_forwardWhat is the condensed structural formula for: A) 2-methyl-2-heptanol B) 3-phenyl-1butanol C) 3-ethyl-2-pentanolarrow_forward
- What are the functional groups present in the compound? Alcohol and carboxylic acid Alcohol and ketone Alcohol and aldehyde Alcohol and Esterarrow_forwardExplain why carboxylic acids have higher boiling points than other compounds with OH groups?arrow_forwardWhat is the functional isomer of 2-propanol? propanoic acid methoxy ethane propanal propanone Other:arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning




