
Concept explainers
Calculate the gas pressure inside the platinum capsule after
Answer to Problem 4.35P
Gas pressure is
Explanation of Solution
When Radium-226 decay, it emits a particles each a particle is a helium nucleus with 2 electrons and forms helium gas which produces pressure in a capsule.
226 Ra emits 4 alpha particles per decay immediately after secular equilibrium and there is the formation of the fifth a particle.
The fraction of
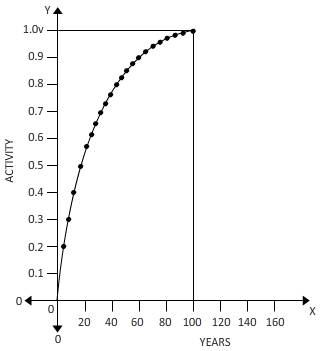
Above carve shows a fraction of a to helium over 100 years.
Fraction=Area under curve/Total area
=
Integrating from
Fraction=
Four alpha particles emitted over
We know that,
To calculate the fraction of decay
Rearranging, (1) and (2)
Hence
To calculate helium atoms formed over
We have,
The dimension of the capsule,
The molecular weight of RaBr2,
The volume of RaBr2 is,
Volume available for gas=volume of capsule-volume of RaBr2
According to Ideal gas low
The pressure of helium formed by the decay of radium is 22.3 atmospheres. But originally capsule contains an air of 1 atmospheric pressure. Hence this initial pressure must be considered.
Thus, pressure=
Conclusion:
We can conclude about this problem that, when Radium-226 decays. It emits a particles having helium nucleus with two electrons and produces helium gas. Ideal gas law is used to calculate the pressure of helium gas. The gas pressure inside the capsule is 23.3 atm due to the formation of helium gas by the decay of radium.
Want to see more full solutions like this?
Chapter 4 Solutions
Introduction To Health Physics
- What is the dose in mSv for: (a) a 0.1 Gy x-ray?(b) 2.5 mGy of neutron exposure to the eye? (c) 1.5 mGy of α exposure?arrow_forwardWhat would the BEN in MeV be for a nucleus with 94 protons, 149 neutrons and an atomic mass of 223.1 u?arrow_forwardWhat is the SI unit for radiation dose? What is its value in J/kg and rad?arrow_forward
- If the energy in fusion bombs were used to supply the energy needs of the world, how many of the 9-megaton variety would be needed for a year’s supply of energy (using data from U(x) = 1/2(kx2) + const.)?arrow_forwardWhat is the mass of 60 Co in a cancer therapy transillumination unit containing 5.00 kCi of 60 Co ?arrow_forwardCalculate the dose in Sv to the chest at a patient given an xray under the following conditions. The xray beam intensity is 1.50 W/m2, the area of the chest exposed is 0.0750 m2 35.0% of the xrays are absorbed in 20.0 kg of tissue, and the exposure time is 0.250 s.arrow_forward
- (a) Show that if you assume the average nucleus is spherical with a radius r=r0A1/3, and with a mass at A u, then its density is independent at A. (b) Calculate that density in u/fm3 and kg/m3, and compare your results with those found in Example 31.1 for 56Fe.arrow_forwardFind the radiation dose in Gy for: (a) A 10-mSv fluoroscopic X-ray series, (b) 50 mSv of skin exposure by an a emitter, (c) 160 mSv of and rays from the 40K in your body.arrow_forward
 Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning
Modern PhysicsPhysicsISBN:9781111794378Author:Raymond A. Serway, Clement J. Moses, Curt A. MoyerPublisher:Cengage Learning College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning
College PhysicsPhysicsISBN:9781285737027Author:Raymond A. Serway, Chris VuillePublisher:Cengage Learning College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College
College PhysicsPhysicsISBN:9781938168000Author:Paul Peter Urone, Roger HinrichsPublisher:OpenStax College Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning
Principles of Physics: A Calculus-Based TextPhysicsISBN:9781133104261Author:Raymond A. Serway, John W. JewettPublisher:Cengage Learning Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning
Classical Dynamics of Particles and SystemsPhysicsISBN:9780534408961Author:Stephen T. Thornton, Jerry B. MarionPublisher:Cengage Learning University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax
University Physics Volume 3PhysicsISBN:9781938168185Author:William Moebs, Jeff SannyPublisher:OpenStax





