
Concept explainers
(a)
Interpretation:
The chemical formula of red compound in the following periodic table is to be identified.
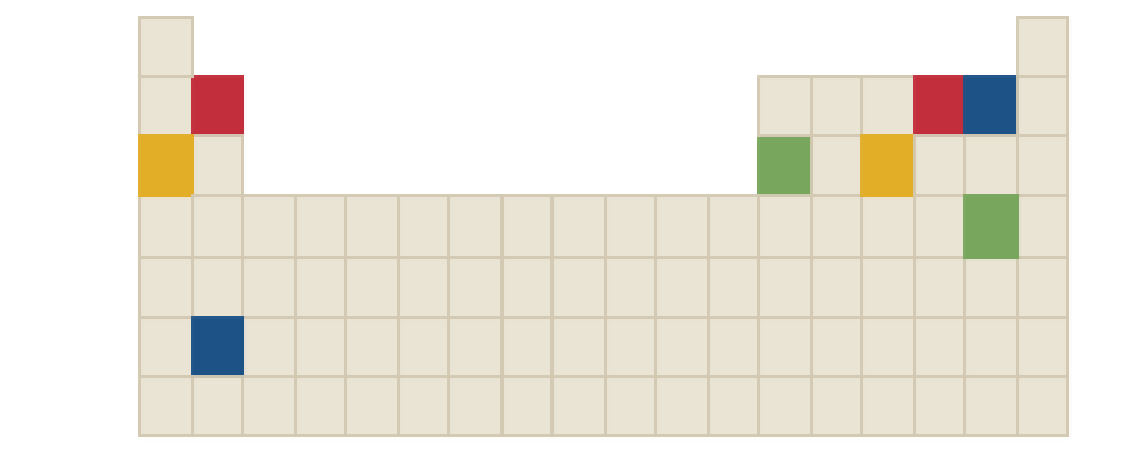
Concept Introduction:
Ions are formed by the loss or gain of electrons. The formation of ion requires the presence of two elements, these two elements are: one is metal atom and another one is non-metal atom. Metal atom loses electron and non-metal atom accepts electron.
The ratio in which positive and negative ions combine is the ratio which achieves charge neutrality for the resulting compound.
There three rules to remember while writing the chemical formulas. They are as follows:
- First write the symbol for positive ion.
- The charges of the ions are not shown in the formula.
- The numbers in the formula give the combining ratio for the ions.
(b)
Interpretation:
The chemical formula of blue compound in the following periodic table is to be identified.
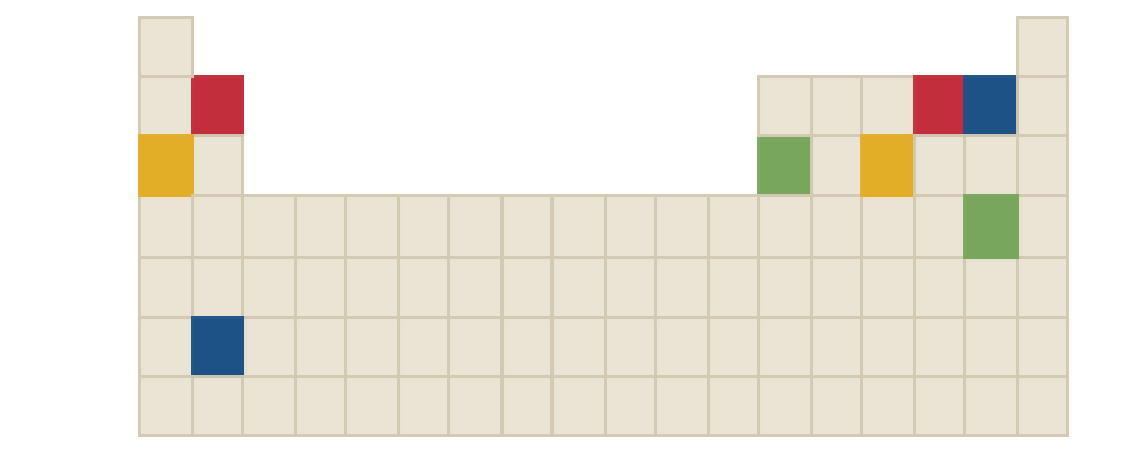
Concept Introduction:
Ions are formed by the loss or gain of electrons. The formation of ion requires the presence of two elements, these two elements are: one is metal atom and another one is non-metal atom. Metal atom loses electron and non-metal atom accepts electron.
The ratio in which positive and negative ions combine is the ratio which achieves charge neutrality for the resulting compound.
There three rules to remember while writing the chemical formulas. They are as follows:
- First write the symbol for positive ion.
- The charges of the ions are not shown in the formula.
- The numbers in the formula give the combining ratio for the ions.
(c)
Interpretation:
The chemical formula of yellow compound in the following periodic table is to be identified.
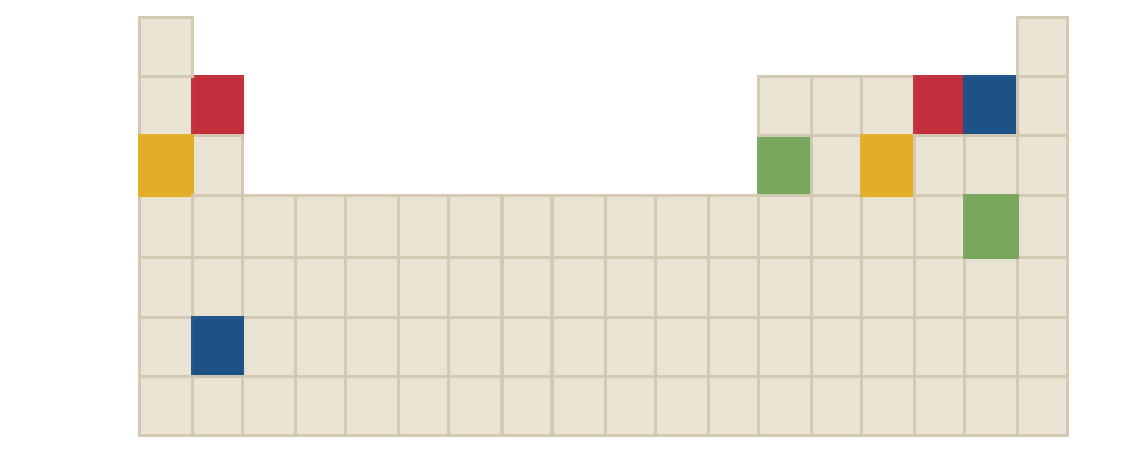
Concept Introduction:
Ions are formed by the loss or gain of electrons. The formation of ion requires the presence of two elements, these two elements are: one is metal atom and another one is non-metal atom. Metal atom loses electron and non-metal atom accepts electron.
The ratio in which positive and negative ions combine is the ratio which achieves charge neutrality for the resulting compound.
There three rules to remember while writing the chemical formulas. They are as follows:
- First write the symbol for positive ion.
- The charges of the ions are not shown in the formula.
- The numbers in the formula give the combining ratio for the ions.
(d)
Interpretation:
The chemical formula of green compound in the following periodic table is to be identified.
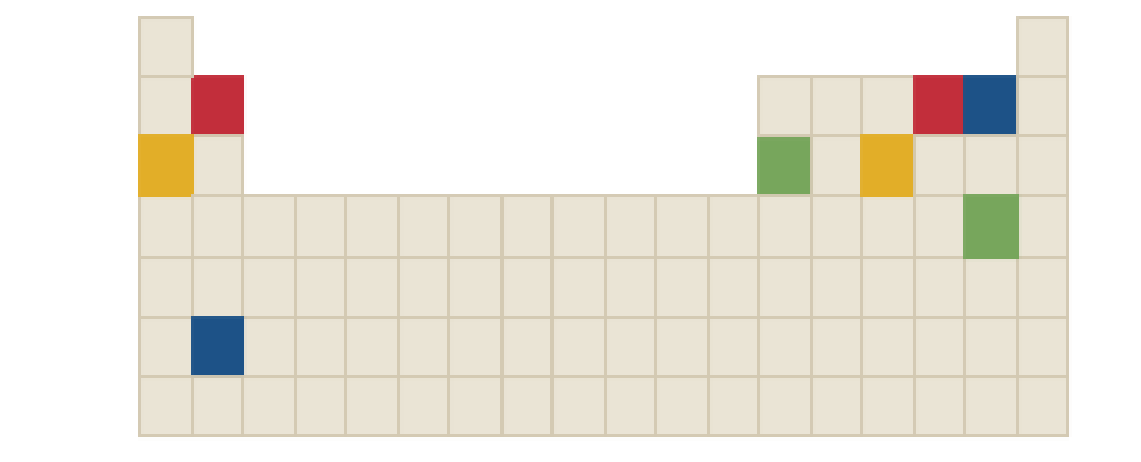
Concept Introduction:
Ions are formed by the loss or gain of electrons. The formation of ion requires the presence of two elements, these two elements are: one is metal atom and another one is non-metal atom. Metal atom loses electron and non-metal atom accepts electron.
The ratio in which positive and negative ions combine is the ratio which achieves charge neutrality for the resulting compound.
There three rules to remember while writing the chemical formulas. They are as follows:
- First write the symbol for positive ion.
- The charges of the ions are not shown in the formula.
- The numbers in the formula give the combining ratio for the ions.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
General, Organic, and Biological Chemistry
- The geometric arrangement of electron pairs around a given atom is determined principal by the tendency to minimize between the electron pairs.arrow_forwardWhich simple ion would each of the following elements he expected to form? What noble gas has an analogous electron configuration to each of the inns? msp;a.chlorine,Z=17b.strontium,Z=38c.oxygen,Z=8d.rubidium,Z=37arrow_forwardIn forming an ionic bond with an atom of chlorine, a sodium atom will: a.receive one electron from the chlorine atom. b.receive two electrons from the chlorine atom. c.give up one electron to the chlorine atom. d.give up two electrons to the chlorine atom.arrow_forward
- Using Lewis Structures to Determine the Correct Chemical Formula for Ionic Compounds Use Lewis structures to determine the correct chemical formula for the compound formed between LiandO.arrow_forwardElement First Ionization Energy (kJImol) Second Ionization Energy (kiImol) I K 419 3050 Ca 590 1140 75. Use principles of atomic structure to answer each of the following: (a) The radius of the Ca atom is 197 pm; the radius of the Ca2+ ion is 99 pm. Account for the difference. (b) The lattice energy of CaO(s) is 3460 kJ/mol; the lattice energy of K2O is 2240 kJ/mol. Account for the difference. (c) Given these ionization values, explain the difference between Ca and K with regard to their first and second ionization energies. Element First Ionization Energy (kJ/mol) Second Ionization Energy (kJ/mol) K 419 3050 Ca 590 1140 (d) The first ionization energy of Mg is 738 kJ/mol and that of Al is 578 kJ/mol. Account for this difference.arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning





