
Concept explainers
(a)
Interpretation:
The name and identification of all the present groups or substituents should be determined in the following compound.
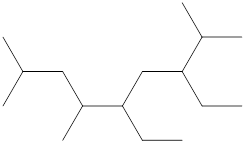
Concept Introduction:
Compounds consist of carbon and hydrogen known as hydrocarbons. Hydrocarbons are classified as a saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which a carbon-carbon single bond is present as carbon is linked with four atoms.
(b)
Interpretation:
The name and identification of all the present groups or substituents should be determined in the following compound.
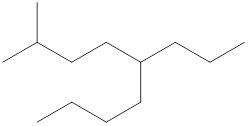
Concept Introduction:
Compounds consist of carbon and hydrogen known as hydrocarbons. Hydrocarbons are classified as a saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which a carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that are double and triple bonds.
(c)
Interpretation:
The name and identification of all the present groups or substituents should be determined in the following compound.
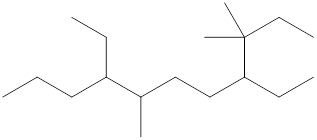
Concept Introduction:
Compounds consist of carbon and hydrogen known as hydrocarbons. Hydrocarbons are classified as a saturated hydrocarbon and unsaturated hydrocarbon. Saturated hydrocarbons are those hydrocarbons in which a carbon-carbon single bond is present as carbon is linked with four atoms. Unsaturated hydrocarbons are those hydrocarbons in which carbon-carbon multiple bonds are present that are double and triple bonds.
Want to see the full answer?
Check out a sample textbook solution
Chapter 4 Solutions
ORGANIC CHEMISTRY (LL)-W/WILEYPLUS
- Distinguish between isomerism and resonance. Distinguish between structural and geometric isomerism. When writing the various structural isomers, the most difficult task is identifying which are different isomers and which are identical to a previously written structurethat is, which are compounds that differ only by the rotation of a carbon single bond. How do you distinguish between structural isomers and those that are identical? Alkenes and cycloalkanes are structural isomers of each other. Give an example of each using C4H8. Another common feature of alkenes and cycloalkanes is that both have restricted rotation about one or more bonds in the compound, so both can exhibit cis- trans isomerism. What is required for an alkene or cycloalkane to exhibit cis-trans isomerism? Explain the difference between cis and trans isomers. Alcohols and ethers are structural isomers of each other, as are aldehydes and ketones. Give an example of each to illustrate. Which functional group in Table 21-4 can be structural isomers of carboxylic acids? What is optical isomerism? What do you look for to determine whether an organic compound exhibits optical isomerism? 1-Bromo-1-chloroethane is optically active whereas 1-bromo-2-chloroethane is not optically active. Explain.arrow_forwardWhat is the relationship, if any, between these organic compounds?arrow_forwardName the cycloalkanes with molecular formula C6H12 that have a 3-membered ring and two substituents.arrow_forward
- Isooctane is the common name of the isomer of C8H18 used as the standard of 100 for the gasoline octane rating: (a) What is the IUPAC name for the compound?(b) Name the other isomers that contain a five-carbon chain with three methyl substituents.arrow_forwardFive structural isomers, or constitutional isomers, have the formula C6H14C6H14. Draw the indicated isomers, grouped by number of carbon atoms in the main chain. a) Draw two isomers with 5 carbon atoms in the main chain. Be sure to include all hydrogen atoms. b) Draw two isomers with 4 carbon atoms in the main chain. Be sure to include all hydrogen atoms.arrow_forwardCyclopentane has a higher boiling point than pentane even though both compounds have the same number of carbons (49 °C vs. 36 °C). Can you suggest a reason for this phenomenon?arrow_forward
- What is the IUPAC name for the compound below? CH2CH3 CH;CHCHCH,CH3 ČH3 O 2-ethyl-3-methylpentane O 4-ethyl-3-methylpentane octane O 2,3-diethylbutane O 3,4-dimethylhexanearrow_forwardIs this correct way of doing it?arrow_forward2,2,4-trimethylpentane is the name of the 100 octane rating in gasoline. Select this molecule and any isomer(s) that contain a five-carbon chain and three methyl substituents (branches).arrow_forward
- Teas fill PDF UNOFFICI TRANSCR 4°F sunny Give the IUPAC name of the following compound: CH3-CH-C=C-CH3 CH₂ CH3 (Hint: Notice that the longest continuous chain of this molecule possesses 6 carbon atoms. You may first want to redraw the formula to have the longest carbon chain in one straight line to facilitate naming.) O 4-ethyl-2-pentene 4-methyl-2-hexyne O 4-ethyl-2-pentyne O 2-ethyl-3-pentyne PDF PDF PDF aster apter 11 emistr... PDF king out pression PDF ab 10 accines per work unizatio ecord PDF C CH 3:3 Bala Top qu 4 Top Questicarrow_forward2. Name each of the following functional Groups. O H H R-O-OH REOH RECI RO-CH₂_R_C=CH H H-CH H R-C-0-CH3 R-O-CH₂ ROH RO-O-CH₂arrow_forwardQ1Carrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning





