
Concept explainers
Interpretation: The reason due to which the products in the cracking of dicyclopentadieneare distilled very slowly needs to be explained.
Concept Introduction:
Dicyclopentadiene has chemical formula C10H12. It is clear yellow color liquid with acrid odor. It is used in resins and also used in adhesives, pants and inks.
It is abbreviated as DCPD and the structure is represented as follows:
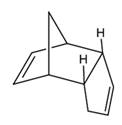
Answer to Problem 1Q
The products are heated slowly to avoid the formation of dicyclopentadiene again.
Explanation of Solution
In the cracking of dicyclopentadiene, the products formed are as follows:
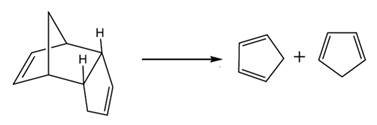
If the products heated strongly, both the products react to form the cyclic reactant again and following reaction take place.
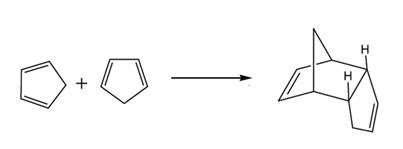
Or,
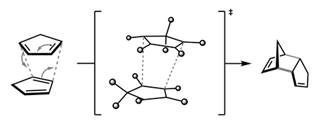
Thus, to avoid the reverse reaction, products are heated slowly.
Want to see more full solutions like this?
Chapter 48 Solutions
Macroscale and Microscale Organic Experiments
- What is the first step of the reaction between 2-methyl cyclohexene and borane in Tetrahydrofuran? Draw the structure of the intermediate.arrow_forwardA Diels–Alder reaction calls for the use of 5.7 mL5.7 mL of a 4.0 M solution of cyclopentadiene in methanol. Calculate the number of moles of cyclopentadiene present in this volume. number of moles of cyclopentadiene: molarrow_forwardThe following table indicates the yield percent values of nitration products and toluene bromination. Explain the difference between the performance percentages of the products of bromination.arrow_forward
- Question: How do quantum mechanical effects influence the stability and reactivity of molecules with non-classical carbocations, such as the 2-norbornyl cation, and how does this impact the reaction mechanisms and outcomes?arrow_forwardThe Diels–Alder Reaction – A [4+2] Cycloaddition Experiment. (a) Explain why the endo-rule is not obeyed when furan is used as the diene. Use chemical structures and models to support your answer.arrow_forwardList the percentages of the products from a single radical bromination of pentane.arrow_forward
- What is the major difference between catalytic reduction and non-catalylic reduction techniques?arrow_forwardWhat diene and dienophile are needed to prepare attached Diels–Alder product?arrow_forwardSynthesis of vinyl acetate from ethylene, acetic acid and oxygen over a Palladium catalyst Why is this the best way to produce vinyl acetate? mention 6 reasons for that ( in points)arrow_forward
- 1. Write down all the steps of the reaction of 2-Methyl-2-hexene with sulfuric acid and name the product formed. Will provide helpful ratings for correct solution.arrow_forwardWhat is the structure of the Diels Alder product according to this reaction?arrow_forwardWhy does a Birch reduction require ROH as the proton source, whereas a dissolving metal reduction of an alkyne uses NH3 as the proton source? Hint: Which acid is stronger?arrow_forward
 Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole
Macroscale and Microscale Organic ExperimentsChemistryISBN:9781305577190Author:Kenneth L. Williamson, Katherine M. MastersPublisher:Brooks Cole

