
Concept explainers
Interpretation:
The difference between alpha and beta glucose is to be found out.
Concept introduction:
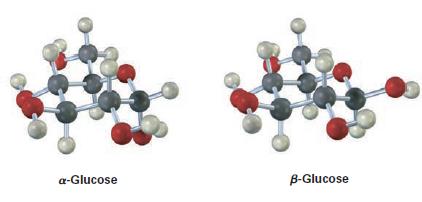
Glucose can form either five-membered or six-membered rings. The six-membered ring is much more common, and in solution the vast majority of glucose molecules are found to have six-membered rings.
Each molecule of glucose has a carbon backbone with -OH groups and hydrogen atoms attached to it. At the top of the chain, an oxygen atom is double-bonded to a carbon atom; collectively, these two atoms are called a carbonyl group. The carbon backbone of the glucose molecule can coil up so that an -OH group near the bottom end of the chain attacks the carbonyl carbon and the glucose molecule forms a ring. This ring-shaped structure is the cyclic form of glucose, while the straight chain structure is the linear form.
Trending nowThis is a popular solution!

Chapter 4 Solutions
Organic Chemistry
- Treatment of -D-glucose with methanol in the presence of an acid catalyst converts it into a mixture of two compounds called methyl glucosides (Section 25.3A). In these representations, the six-membered rings are drawn as planar hexagons. (a) Propose a mechanism for this conversion and account for the fact that only the OH on carbon 1 is transformed into an OCH3 group. (b) Draw the more stable chair conformation for each product. (c) Which of the two products has the chair conformation of greater stability? Explain.arrow_forwardAll the glucose units in dextran have six-membered rings. When a sample of dextran is treated with methyl iodide and Ag2O and the product ishydrolyzed under acidic conditions, the final products are 2,3,4,6-tetra-O-methyl-d-glucose, 2,4,6-tri-O-methyl-d-glucose, 2,3,4-tri-O-methyl-d-glucose, and 2,4-di-O-methyl-d-glucose. Draw a short segment of dextran.arrow_forward1. Show the steps in drawing the alpha pyranose of D-sorbose. 2. Show the steps in drawing the boat conformation of D-sorbose. 3. Show the steps in drawing the chair conformation of D-sorbose.arrow_forward
- In addition to more highly fluorinated products, fluorination of 2-methylbutane yields a mixture of compounds with the formula C5H10F2. Draw the structures of all the isomers with the formula C5H10F2 that would be produced and label with a star all the chiral centers present in their structures.arrow_forwardThe structure of 4 isomers of ketopentose are shown. 1) select every structure that is a diastereomer of structure D A, B, or C? 2) select every structure that is a enantiomer of structure C A, B, or D 3) select every structure that is a stereoisomer of structure C A, B, or Darrow_forwardD-glucose and L-glucose would be expected to show differences in which of the followingp roperties? (Answer the items as dif erent or not different) a. Solubility in an achiral solvent b. Density c. Melting point d. Solubility in a chiral solvent e. Freezing point f. Reaction with ethanol (achiral compound) g. Reaction with (+)-lactic acid (chiral compound)arrow_forward
- Rank the following groups in order of decreasing priority. −Cl, −CH3, −SH, −OHarrow_forwardThe structure of 4 isomers of an aldotetrose carbohydrate are given. 1) select every structure that is a diastereomer of structure D A, B, or C? 2) select every structure that is a enantiomer of structure C D, B, or A? 3) select every structure that is a stereoisomer of structure D A, B, or Carrow_forwardDraw the highest and lowest energy conformations. In cases where two or three conformations are degenrate, choose only one as your answer. Use Ch2Ch3, H and Ch3 for the structure.arrow_forward

 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


