
Concept explainers
You have seven closed containers, each with equal masses of chlorine gas (Cl2) . You add 10.0 g of sodium to the first sample, 20.0 g of sodium to the second sample, and so on (adding 70.0 g of sodium to the seventh sample). Sodium and chlorine react to form sodium chloride according to the equation
After each reaction is complete, you collect and measure the amount of sodium chloride formed. A graph of your results is shown below.
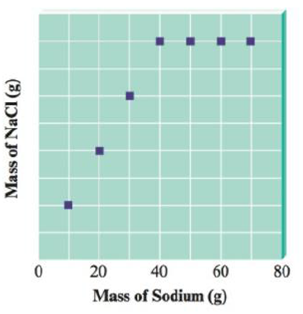
Answer the following questions:
a. Explain the shape of the graph.
b. Calculate the mass of NaCl formed when 20.0 g of sodium is used.
c. Calculate the mass of Cl2 in each container.
d. Calculate the mass of NaCl formed when 50.0 g of sodium is used.
e. Identify the leftover reactant, and determine its mass for parts b and d above.
(a)
Interpretation: Seven closed containers, having equal amount of chlorine gas present in them, have been given. Sodium is added to the containers in the given pattern. After the completion of the reaction between sodium and chlorine, the amount of sodium chloride formed is measured. The stated questions related to the given process are to be answered.
Concept introduction: The amount of the sodium chloride formed depends upon the amount of sodium and chlorine gas present in each of the given containers.
To determine: The explanation regarding the shape of the graph.
Explanation of Solution
The amount of sodium chloride formed increases on increasing the quantity of sodium from the container one to four.
In the fifth container the addition of a greater amount of sodium than added to the previous container did not increase the amount of sodium chloride formed.
This indicates that the amount of chlorine gas present in the respective container is not sufficient to react with amount of sodium been added to the container. Hence, the amount of sodium chloride formed in the subsequent containers has a constant value.
The amount of chlorine gas present in the containers five, six and seven reacts with an equal amount of sodium to form an equal amount of sodium chloride.
(b)
Interpretation: Seven closed containers, having equal amount of chlorine gas present in them, have been given. Sodium is added to the containers in the given pattern. After the completion of the reaction between sodium and chlorine, the amount of sodium chloride formed is measured. The stated questions related to the given process are to be answered.
Concept introduction: The amount of the sodium chloride formed depends upon the amount of sodium and chlorine gas present in each of the given containers.
To determine: The mass of
Explanation of Solution
Given
The stated reaction is,
The atomic mass of sodium is
The molar mass of
According to the given reaction,
The mass of
(c)
Interpretation: Seven closed containers, having equal amount of chlorine gas present in them, have been given. Sodium is added to the containers in the given pattern. After the completion of the reaction between sodium and chlorine, the amount of sodium chloride formed is measured. The stated questions related to the given process are to be answered.
Concept introduction: The amount of the sodium chloride formed depends upon the amount of sodium and chlorine gas present in each of the given containers.
To determine: The mass of
Explanation of Solution
Given
The stated reaction is,
The atomic mass of sodium is
The molar mass of
According to the given graph, chlorine gas present reacts completely with
According to the given reaction,
The mass of
(d)
Interpretation: Seven closed containers, having equal amount of chlorine gas present in them, have been given. Sodium is added to the containers in the given pattern. After the completion of the reaction between sodium and chlorine, the amount of sodium chloride formed is measured. The stated questions related to the given process are to be answered.
Concept introduction: The amount of the sodium chloride formed depends upon the amount of sodium and chlorine gas present in each of the given containers.
To determine: The mass of
Explanation of Solution
Given
The stated reaction is,
The atomic mass of sodium is
The molar mass of
According to the given graph, chlorine gas present reacts completely with
Hence, only
According to the given reaction,
The mass of
(e)
Interpretation: Seven closed containers, having equal amount of chlorine gas present in them, have been given. Sodium is added to the containers in the given pattern. After the completion of the reaction between sodium and chlorine, the amount of sodium chloride formed is measured. The stated questions related to the given process are to be answered.
Concept introduction: The amount of the sodium chloride formed depends upon the amount of sodium and chlorine gas present in each of the given containers.
To determine: The left over reactant in parts (b) and (d) and their respective mass.
Explanation of Solution
The leftover reactant in part (b) is the chlorine gas. The mass of chlorine left unreacted is
To determine: The left over reactant in parts (b) and its respective mass.
The leftover reactant in part (b) is the chlorine gas. The mass of chlorine left unreacted is
Given
The stated reaction is,
The atomic mass of sodium is
The molar mass of
According to the given reaction,
The mass of chlorine present in the container is
The amount of chlorine left unreacted is
To determine: The left over reactant in parts (d) and its respective mass.
The leftover reactant in part (d) is sodium. The mass of sodium left unreacted is
According to the given graph, chlorine gas present reacts completely with
Hence, only
The amount of sodium left unreacted is
The mass of chlorine gas left unreacted in the stated reaction
The mass of sodium left unreacted in the stated reaction
Want to see more full solutions like this?
Chapter 5 Solutions
Chemistry: An Atoms First Approach
- Two general chemistry students working together in the lab weigh out 0.832 g of CaCl2 2 H2O into a crucible. After heating the sample for a short time and allowing the crucible to cool, the students determine that the sample has a mass of 0.739 g. They then do a quick calculation. On the basis of this calculation, what should they do next? (a) Congratulate themselves on a job well done. (b) Assume the bottle of CaCl2 2 H2O was mislabeled; it actually contained something different. (c) Heat the crucible again, and then reweigh it.arrow_forwardCopper reacts with nitric acid according to the following reaction. 3Cu(s)+8HNO3(aq)3Cu(NO3)2(aq)+2NO(g)+4H2O(l) If 2.40 g of Cu is added to a container with 2.00 mL of concentrated nitric acid (70% by mass HNO3; density = 1.42 g/cm3), what mass of nitrogen monoxide gas will be produced?arrow_forwardWhen washing soda, Na2CO3, reacts with sulfuric acid, H2SO4, the equation is Na2CO3(aq) + H2SO4(aq) CO2(g) + H2O() + Na2SO4(aq) (a) Describe in words the meaning of this chemical equation. (b) Verify that the equation conforms to the law of conservation of matter.arrow_forward
- When the supply of oxygen is limited, iron metal reacts with oxygen to produce a mixture of FeO and Fe2O3. In a certain experiment, 20.00 g iron metal was reacted with 11.20 g oxygen gas. After the experiment, the iron was totally consumed, and 3.24 g oxygen gas remained. Calculate the amounts of FeO and Fe2O3 formed in this experiment.arrow_forwardConsider the chemical reaction 2 S + 3 O2 → 2 SO3. If the reaction is run by adding S indefinitely to a fixed amount of O2, which of these graphs best represents the formation of SO3? Explain your choice.arrow_forwardThe reaction of iron metal and chlorine gas to give iron(III) chloride is illustrated below. (a) Write the balanced chemical equation for the reaction. (b) Beginning with 10.0 g of iron, what mass of Cl2, in grams, is required for complete reaction? What mass of FeCl3 can be produced? (c) If only 18.5 g of FeCl3 is obtained from 10.0 g of iron and excess Cl2, what is the percent yield? (d) If 10.0 g each of iron and chlorine are combined, what is the theoretical yield of iron(III) chloride?arrow_forward
- 3.83 For the reaction of nitrogen, N2, and hydrogen, H2, to form ammonia, NH3, a student is attempting to draw a particulate diagram, as shown below. Did the student draw a correct representation of the reaction? If not, what was the error the student made?arrow_forward4.69 The pictures below show a molecular-scale view of a chemical reaction between H2 and CO to produce methanol, CH3OH. The box on the left represents the reactants at the instant of mixing, and the box on the right shows what is left once the reaction has gone to completion. Was there a limiting reactant in this reaction? If so, what was it? Write a balanced chemical equation for this reaction. As usual, your equation should use the smallest possible whole number coefficients for all substances.arrow_forwardYou have a pure sample of apholate, C12H24N9P3, a highly effective commercial insecticide. Calculate the molar mass of apholate. Calculate the mass of N in 100. g apholate. A sample containing 250.0 mg apholate is sprayed on an agricultural field. Calculate the mass of phosphorus in this sample of apholate; express your result in grams. Calculate the number of phosphorus atoms in this sample of apholate.arrow_forward
- 3.113 MgCl2 is often found as an impurity in table salt (NaCl). If a 0.05200-g sample of table salt is found to contain 61.10% Cl by mass, describe how you could determine the percentage of MgCl2 in the sample.arrow_forwardBoron forms an extensive series of compounds with hydrogen, all with the general formula BxHy. To analyze one of these compounds, you burn it in air and isolate the boron in the form of B2O3 and the hydrogen in the form of water. You find that 0.1482 g BxHy gives 0.4221 g B2O3 when burned in excess O2. Determine the empirical I formula of BxHy.arrow_forwardThe sugar sucrose, which is present in many fruits and vegetables, reacts in the presence of certain yeast enzymes to produce ethanol and carbon dioxide gas. Balance the following equation for this reaction of sucrose. C12H22O11(aq) + H2O(l) C2H5OH(aq) + CO2(g)arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





