
(a)
Interpretation:
Condensed structural formula for acetyl chloride has to be drawn.
Concept Introduction:
General structure of acid chloride can be represented as shown below,
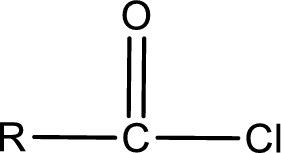
The structure of the acid chloride can be derived from the IUPAC name or common name. “R” group present in the above structure is a part of the parent
(b)
Interpretation:
Condensed structural formula for 2-methylbutanoyl chloride has to be drawn.
Concept Introduction:
General structure of acid chloride can be represented as shown below,
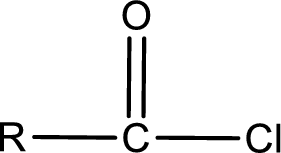
The structure of the acid chloride can be derived from the IUPAC name or common name. “R” group present in the above structure is a part of the parent carboxylic acid from which the acid chloride is obtained. Name of the acid chloride is obtained by replacing “-oic acid” in the parent carboxylic acid name with “-oyl chloride”.
(c)
Interpretation:
Condensed structural formula for propionic anhydride has to be drawn.
Concept Introduction:
General structure of acid anhydride can be represented as shown below,
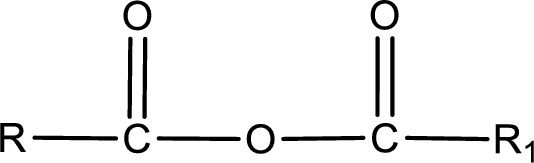
The structure of the acid anhydride can be derived from the IUPAC name or common name. “R, R1” group present in the above structure is a part of the parent carboxylic acid from which the acid anhydride is obtained. If the acid anhydride is a symmetric one, then the name of acid anhydride is given by replacing the acid in the parent carboxylic acid name with anhydride. If the acid anhydride is an asymmetric one, then the name of acid anhydride is given by using the name of individual carboxylic acid in an alphabetical order followed by anhydride.
(d)
Interpretation:
Condensed structural formula for ethanoic methanoic anhydride has to be drawn.
Concept Introduction:
General structure of acid anhydride can be represented as shown below,
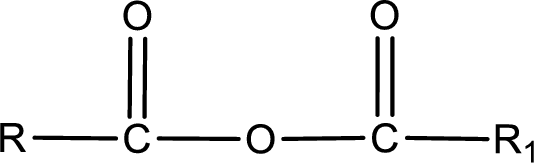
The structure of the acid anhydride can be derived from the IUPAC name or common name. “R, R1” group present in the above structure is a part of the parent carboxylic acid from which the acid anhydride is obtained. If the acid anhydride is a symmetric one, then the name of acid anhydride is given by replacing the acid in the parent carboxylic acid name with anhydride. If the acid anhydride is an mixed one, then the name of acid anhydride is given by using the name of individual carboxylic acid in an alphabetical order followed by anhydride.
Want to see the full answer?
Check out a sample textbook solution
Chapter 5 Solutions
Organic And Biological Chemistry
- An anhydride is an organic molecule derived from two carboxylic acids. What are these two acids for the molecule in the picture? i. Two butanoic acidii. One octanoic acidiii. Butanoic acid + propanoic acidiv. Acetic acid + butanoic acidarrow_forwardPhthalate esters are used as plasticizers. What is a plasticizer? Explain why plastics become brittle over time.arrow_forwardPlease name the following alchol or carboxylic acid/ester (spell out the full name of the compound) :) A) Refer to the first photo for the alcohol B) Refer to the second photo for the carboxylic acid or esterarrow_forward
- What reagents are used in the esterification of Alcohols and Phenols? a. Write the reaction involved in Primary Alcohol (Ethanol) and Acetyl Chloride b. Write the reaction involved in Phenol and Acetyl Chloride What is the purpose of the Chromic acid test? a. What are the reagents used? b. Write oxidation reaction of Primary Alcohols and Secondary Alcoholsarrow_forwarda) indicate the structural formula of the product/s formed b) name the organic product formed c) indicate the type of reaction d) catalyst usedarrow_forwardWhat is the ester found in: Bananas, Oranges, Strawberries, Peach, Apricots, and liniments?arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning




