
Use the molecular art to identify the process as a
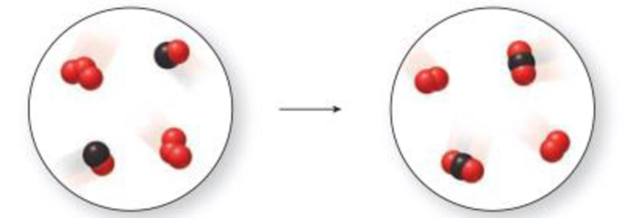
Interpretation:
The type of change (physical or chemical change) represented by the molecular art has to be given and explained. If the process is physical, the phase change has to be written and if the process is chemical, the equation has to be written.
Concept Introduction:
Physical change: An alteration of substance without changing its composition is called as physical change. The change in state of a substance is the most common physical change. Example: Melting of an ice cube to form liquid water.
Chemical change: The conversion of one material to another is called as chemical change (or) chemical reaction. Example: formation of water from hydrogen and oxygen is a chemical reaction because there is difference in the composition of the material at the beginning and end of the process.
Explanation of Solution
The given representation is,
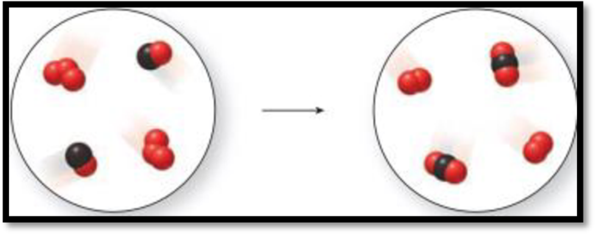
Figure 1
The above represent represents a chemical change because new products are formed by chemical bonds.
The three red spheres represent ozone
The combination of red and black spheres represents carbon monoxide
The combination of two red spheres with one black sphere represents carbon dioxide
The two red spheres represent oxygen
The chemical equation is,
The reaction is not balanced.
The balanced equation is,
Want to see more full solutions like this?
Chapter 5 Solutions
Principles of General, Organic, Biological Chemistry
- Table 6.1 lists some clues that a chemical reaction has occurred. However, these events do not necessarily prove the existence of a chemical change. Give an example for each of the clues that is not a chemical reaction but a physical change.arrow_forwardhy is the formation of a gas evidence of a chemical reaction? Use a molecular-level drawing in your explanation.arrow_forwardAcid rain is rainfall that contains sulfuric acid originating from organic fuels that contain sulfur. The process occurs in three major steps. The sulfur burns first, forming sulfur dioxide. In sunlight, the sulfur dioxide reacts with oxygen in the air to produce sulfur trioxide. When rainwater falls through the sulfur trioxide, the reaction produces sulfuric acid. Write the equation for each step in the process and tell what kind of reaction it is.arrow_forward
- Which of the following statements about chemical reactions is false? l type='a'> When balancing a chemical equation, all subscripts must be conserved. i>When one coefficient is doubled, the rest of the coefficients in (he balanced equation must also be doubled. i>The subscripts in a balanced equation tell us the number of atoms in a molecule. i>The phases in a chemical reaction tell us the nature of the reactants and products.arrow_forwardExplain how an equation can be balanced even if the number of reactant particles differs from the number of product particles.arrow_forwardYou have probably had the unpleasant experience of discovering that a flashlight battery has gotten old and begun to leak. Is there evidence that this change is due to a chemical reaction?arrow_forward
- What is a combustion reaction? Are combustion reactions a unique type of reaction, or are they a special case of a more general type of reaction? Write an equation that illustrates a combustion reaction.arrow_forwardList and define all the ways of classifying chemical reactions that have been discussed in the text. Give a balanced chemical equation as an example of each type of reaction, and show clearly how your example fits the definition you have given.arrow_forwardWhat are the substances to theleftof the arrow in a chemical equation called? To therightof the arrow? What does the arrow itself mean?arrow_forward
 Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning





