
Concept explainers
Without referring to Figure 5.1, write Lewis dot symbols for the following atoms/ions: (a) Be, (b) K, (c) Ca, (d) Ga, (e) O, (f) Li+, (g) Cl–, (h) S2−, (i) Sr2+, (j) N3−.
(a)
Interpretation:
Lewis dot symbol for the given atoms/ions has to be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
Answer to Problem 5.6QP
Lewis dot representation for
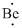
Explanation of Solution
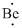
The electronic configuration of Beryllium is
(b)
Interpretation:
Lewis dot symbol for the given atoms/ions has to be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
Answer to Problem 5.6QP
Lewis dot representation for
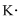
Explanation of Solution
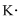
The electronic configuration of Potassium is
(c)
Interpretation:
Lewis dot symbol for the given atoms/ions has to be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
Answer to Problem 5.6QP
Lewis dot representation for
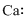
Explanation of Solution
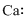
The electronic configuration of Calcium is
(d)
Interpretation:
Lewis dot symbol for the given atoms/ions has to be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
Answer to Problem 5.6QP
Lewis dot representation for
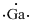
Explanation of Solution
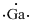
The electronic configuration of Gallium is
(e)
Interpretation:
Lewis dot symbol for the given atoms/ions has to be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
Answer to Problem 5.6QP
Lewis dot representation for
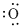
Explanation of Solution
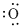
The electronic configuration of oxygen is
Lewis dot symbol denotes the valence electron of an atom. The valence electron of
(f)
Interpretation:
Lewis dot symbol for the given atoms/ions has to be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
Answer to Problem 5.6QP
Lewis dot representation for
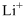
Explanation of Solution
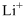
The electronic configuration of Lithium is
(g)
Interpretation:
Lewis dot symbol for the given atoms/ions has to be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
Answer to Problem 5.6QP
Lewis dot representation for
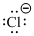
Explanation of Solution
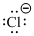
The electronic configuration of Chlorine is
(h)
Interpretation:
Lewis dot symbol for the given atoms/ions has to be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
Answer to Problem 5.6QP
Lewis dot representation for
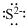
Explanation of Solution
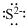
The electronic configuration of Sulphur is
(i)
Interpretation:
Lewis dot symbol for the given atoms/ions has to be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
Answer to Problem 5.6QP
Lewis dot representation for
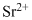
Explanation of Solution
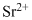
The electronic configuration of Strontium is
(j)
Interpretation:
Lewis dot symbol for the given atoms/ions has to be shown
Concept Introduction:
Lewis dot symbol is used to represent the valence electrons of an atom or ion using dots surrounding the element symbol along four sides of the element symbol without maintaining exact order for the placement of dots.
In Lewis dot symbol representation, the symbol of element is surrounded by "dots" indicating the number of valence electrons available for the element. The dots can be placed one at a time on all the four sides, further electron can be placed by pairing up with the first placed dots. According to the number of electrons added or removed, charge must be placed on the Lewis dot symbol for cations and anions.
The valence electron is the number of electrons present in the outermost shell of the atom. The number of valence electrons will be same for the same group elements which is represented by Lewis dot symbol.
Answer to Problem 5.6QP
Lewis dot representation for
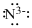
Explanation of Solution
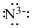
The electronic configuration of Nitrogen is
Want to see more full solutions like this?
Chapter 5 Solutions
Chemistry: Atoms First
- Ca, Cl, Na, Fe, and K. Which of these elements form cations? List their symbols with charges. Which of these elements form anions? List their symbols with charges.arrow_forwardWhich compound of calcium is used for swimming pool water and sewage treatment? (A) Calcium hydroxide, Ca(OH)2 (B) Calcium hypochlorite, Ca(OCl)2 (C) Calcium carbonate, CaCO3 (D) Calcium oxide, CaOarrow_forwardExplaining to me in great detail how to name Sr(OH)2 and Fe(OH)3 compounds. What type of compounds are they? What are the charges on the metals? How are the nomenclature rules different for these two compounds?arrow_forward
- Write the balanced equation for the photosynthesis reaction in which gaseous carbon dioxide and liquid water react in the presence of chlorophyll to produce aqueous glucose (C6H12O6) and oxygen gas.arrow_forwardDistinguish between the terms molecule and ion pairarrow_forwardElements in the same group of the periodic table often form oxyanions with the same general formula. The anions are also named in a similar fashion. Based on these observations, suggest a chemical formula or name, as appropriate, for each of the following ions: (a) BrO4 -, (b) SeO3 2-, (c) arsenate ion, (d) hydrogen tellurate ion.arrow_forward
- Write the symbol for F, Cl, Br, I, O when they react to form ions. Are they cations or anions?arrow_forwardWrite the empirical formula of the compound formed by (a) Al3+ and Cl - ions, (b) Al 3+ and O2 - ions, (c) Mg2+ and NO3 ions.arrow_forwardWrite an equation that shows the formation of a iron(II) ion from a neutral iron atom.Represent electrons as e- .If a box is not needed, leave it blank.If the coefficient is 1, do not write it.arrow_forward
- Explain how the atomic mass unit (amu) is defined and explain how the definition of an amu, in turn, determines the value of Avogadro’s number (NA), which is 6.0221 x 1023arrow_forward3.116 The simplest approximate chemical formula for the human body could be written as C728H4850O1970N104Ca24P16K4S4Na3Cl2Mg. Based on this formula, describe how you would rank by mass the ten most abundant elements in the human body.arrow_forwardhow many grams of lithium nitrate will be needed to make 250 grams of lithium sulfate. assuming that you have an adequate amount of lead (IV) sulfate to do the reaction?arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning





