
Concept explainers
(a)
Interpretation:
Structural formula for ethyl butyrate has to be drawn.
Concept Introduction:
General structure of ester can be represented as shown below,
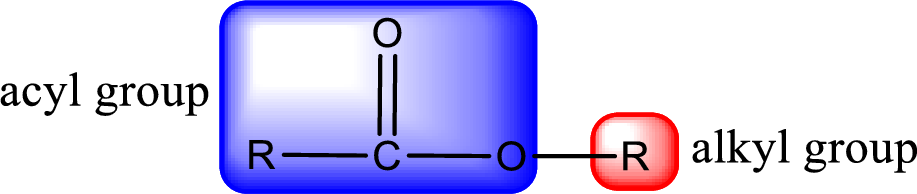
From an IUPAC name, the structure of the ester can be derived. IUPAC name of ester consists of two parts. In an IUPAC name of ester, the first part of the name is the alkyl part and the second part is the acyl part. Alkyl group have come from the alcohol and acyl part from
The same rule applies for deriving a structure from common name. The only difference is the acyl part name. The acyl part is named using the common name of carboxylic acid.
(b)
Interpretation:
Structural formula for butyl ethanoate has to be drawn.
Concept Introduction:
General structure of ester can be represented as shown below,
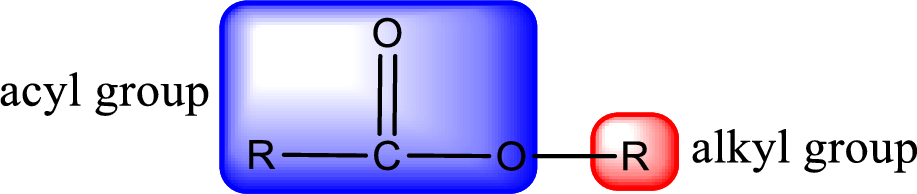
From an IUPAC name, the structure of the ester can be derived. IUPAC name of ester consists of two parts. In an IUPAC name of ester, the first part of the name is the alkyl part and the second part is the acyl part. Alkyl group have come from the alcohol and acyl part from carboxylic acid.
The same rule applies for deriving a structure from common name. The only difference is the acyl part name. The acyl part is named using the common name of carboxylic acid.
(c)
Interpretation:
Structural formula for 2-methylpropyl formate has to be drawn.
Concept Introduction:
General structure of ester can be represented as shown below,
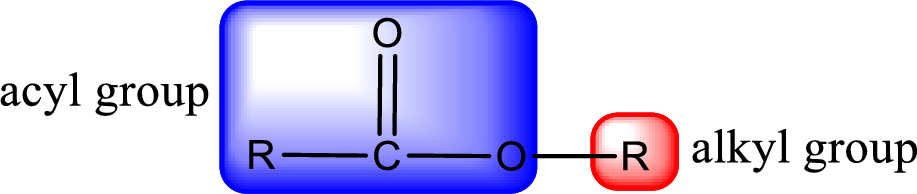
From an IUPAC name, the structure of the ester can be derived. IUPAC name of ester consists of two parts. In an IUPAC name of ester, the first part of the name is the alkyl part and the second part is the acyl part. Alkyl group have come from the alcohol and acyl part from carboxylic acid.
The same rule applies for deriving a structure from common name. The only difference is the acyl part name. The acyl part is named using the common name of carboxylic acid.
(d)
Interpretation:
Structural formula for ethyl
Concept Introduction:
General structure of ester can be represented as shown below,
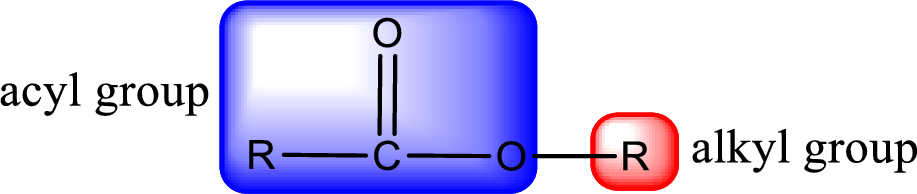
From an IUPAC name, the structure of the ester can be derived. IUPAC name of ester consists of two parts. In an IUPAC name of ester, the first part of the name is the alkyl part and the second part is the acyl part. Alkyl group have come from the alcohol and acyl part from carboxylic acid.
The same rule applies for deriving a structure from common name. The only difference is the acyl part name. The acyl part is named using the common name of carboxylic acid.
Trending nowThis is a popular solution!

Chapter 5 Solutions
Organic And Biological Chemistry
- 8. The organic starting materials for the preparation of an ester could be_________A. a ketone and alcohol B. water and oxygen C. an acid and alcohol D. alkane and aldehydearrow_forwardWhat is a polymer? a. any organic compound made by combining smaller molecules b. any synthetic organic compound c. a long chain of repeating molecular units d. esters with a triglyceride structurearrow_forwardGive the structural formulas for:(i) Methyl Ethanoate(ii) Ethyl ethanoateWrite two uses of Ester?arrow_forward
- draw the condensed formulas for the carboxylic acid and alcohol that form these esters:arrow_forwardDraft Two structural features determine the chemistry and properties of aldehydes and ketones ?arrow_forwardIdentify the Classification of Hazardous Wastes. example: Dichloromethane - Halogenated organic solvent (a) Benzoic acid (b) Cellulose (c) Caffeine (d) Ethanolarrow_forward
- explain the structures and properties of functional group :carboxylic acid and aminoarrow_forwardWhat kind of reactions are common to carboxylic acids and esters? Provide an example of each.arrow_forwardExplain what is meant by oxidation in organic moleculesand provide an example of an oxidation reaction involving an aldehydearrow_forward
- What are the generic structures for carboxylic acids and esters? Write a structure for a specific carboxylic acid and ester.arrow_forwarda)why can esterification also be considered to be a neutralization reaction? b)What is distillation? How would distillation be used to purify an ester?arrow_forwardWhat are synthetic polymers?arrow_forward
 Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781285199030Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHERChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHERChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





