
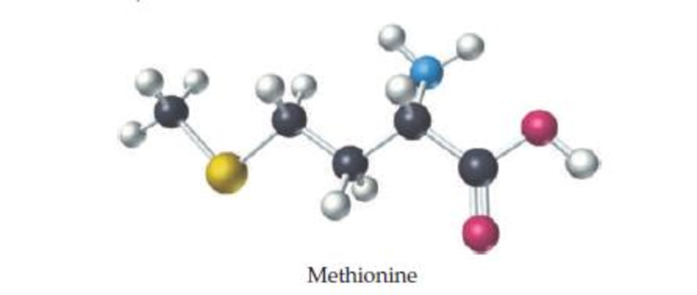
Methionine, an amino acid used by organisms to make proteins, can be represented by the following ball-and-stick molecular model. Write the formula for methionine, and give its molecular weight (red = O, black = C, blue = N, yellow = S, white = H).
Interpretation:
The formula and molecular weight for methionine and should be determined by using the given ball- and- stick molecular model.
Concept Introduction:
Molecular Formula: It is the simplest representation of composition of chemical substance where each and every type of atom is written with the total number of the atom present in the substance.
Molecular weight: The sum of mass of all atoms present in formula of chemical substance is referred as molecular or formula weight of that substance.
Answer to Problem 6.15UKC
The empirical formula for the given structure is
The molecular weight of
Explanation of Solution
Comparing the given details such as red color indicates
The molecular weight for
Finally, molecular weight is obtained by adding
The molecular formula for methionine and molecular weight is determined by using the given ball- and- stick molecular model with details.
Want to see more full solutions like this?
Chapter 6 Solutions
Modified Masteringchemistry With Pearson Etext -- Valuepack Access Card -- For Fundamentals Of General, Organic, And Biological Chemistry
- Glutathione, a powerful antioxidant that destroys harmful oxidizing agents in cells, is composed of glutamic acid, cysteine, and glycine, and has the following structure. a.) What product is formed when glutathione reacts with an oxidizing agent?b.) What is unusual about the peptide bond between glutamic acid and cysteine?arrow_forwardDraw condensed structural formulas for the two carboxylic acids with the molecular formula C4H8O2arrow_forwardWhat is the “octet rule” and its biologically important exception?arrow_forward
- Consider the following acids and their ionization constant, determine which conjugate base is HCOOH Ka = 1.7 x 10-4 (b) HCN Ka = 4.9 x 10-10arrow_forwardV and K are constants that are > 0, explain why the units of K and V are μm and μm/s respectively?arrow_forwardWrite the chemical formula for the conjugate base of each of the following acids: Carbonic Acid (H2CO3)___________________ Formic Acid (CH2O2)_____________________arrow_forward
- Which of the following compounds are acids, and which are bases? Explain.arrow_forwardWrite the chemical formula of the conjugate base of boric acid.arrow_forwardIdentify the conjugate acid-base pairs in the following reactions: HNO2(aq) + H2O(l) → NO2 – (aq) + H3O+(aq) _______ ______ _________ ________ CH3NH2 + H2O(l) → CH3NH3+ + OH – _______ ________ ________ _________arrow_forward
- Which of the following structures exhibit geometric isomerism? Draw and name the two in each case.arrow_forwardIt is speculated that, in 1954, the scientist Allan Turing (who was instrumental in decoding the German Enigma code during the Second World War) ate an apple, which had been injected with a solution of cyanide, to commit suicide. How does cyanide poisoning work on a molecular level, and why can this type of toxic effect not be prevented as in the case of methanol poisoning?arrow_forwardFor in the solution part for c), do you mean 6.25%?arrow_forward
 Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning
Principles Of Radiographic Imaging: An Art And A ...Health & NutritionISBN:9781337711067Author:Richard R. Carlton, Arlene M. Adler, Vesna BalacPublisher:Cengage Learning

