
Organic Chemistry
8th Edition
ISBN: 9781305580350
Author: William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Question
Chapter 6, Problem 6.49P
Interpretation Introduction
Interpretation: The differences for stability of two given carbocations that is
Concept introduction:
Carbocation: The carbon ion that bears a positive charge on it.
Carbocation stability order:
The general carbocation stability order is as follows,
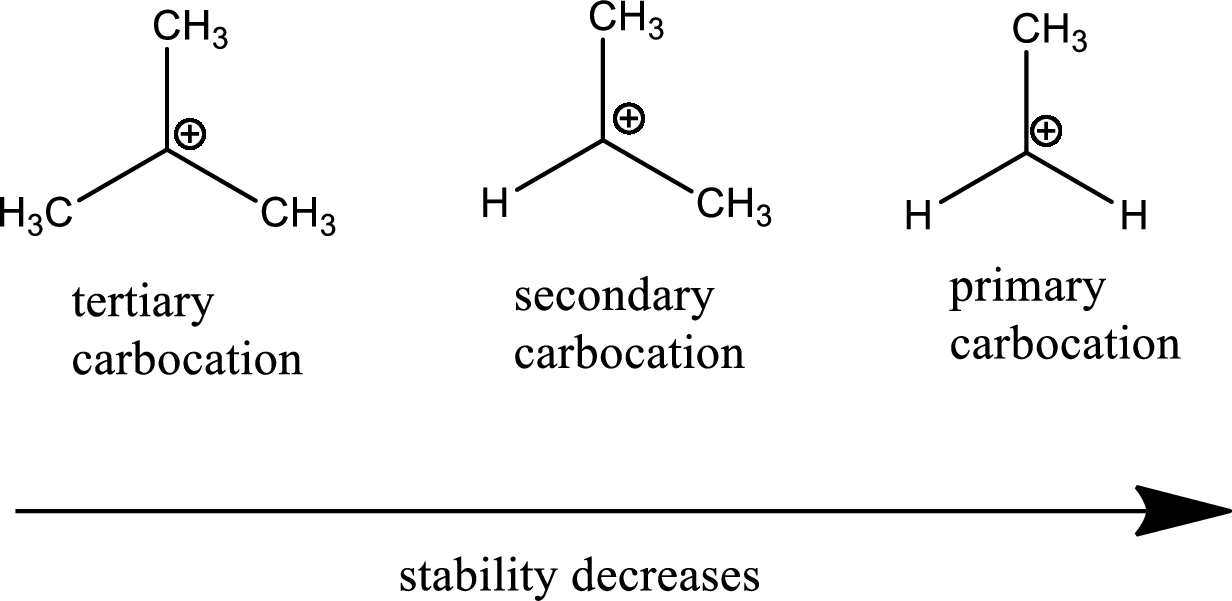
Resonance stabilization: Due to the delocalization of electrons within the molecule the overall energy becomes lower and makes that molecule more stable.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
Carbocations undergo 1,2-methyl shift or 1,2-hydride shift to produce a more stable intermediate. Give the major product(s) obtained from the reaction of the alkene with HBr.SHOW THE FORMATION OF THE CARBOCATION, THE SHIFT THAT MAY OCCUR, AND THE MAJOR PRODUCT FORMED.
What would the major organic reaction product be from the reaction of 1-bromo-1-methylcyclopentane withsodium hydroxide? Would the elimination reaction outcome be affected if a student accidentally adds sodium tertbutoxide instead of sodium hydroxide?
Given that a primary carbocation is less stable than a tertiary carbocation, would you expect that a reaction that creates a primary carbocation would be more or less responsive to solvent polarity than a reaction that creates a tertiary carbocation, and why?
Chapter 6 Solutions
Organic Chemistry
Ch. 6.2 - Using the BDE values from Appendix 3, calculate...Ch. 6.3 - Name and draw a structural formula for the product...Ch. 6.3 - Prob. 6.3PCh. 6.3 - Propose a mechanism for the addition of HI to...Ch. 6.3 - Prob. 6.5PCh. 6.3 - Propose a mechanism for the acid-catalyzed...Ch. 6.3 - The acid-catalyzed hydration of...Ch. 6.3 - Complete these reactions. (a) (b)Ch. 6.3 - Draw the structure of the chlorohydrin formed by...Ch. 6.4 - Draw structural formulas for the alkene that gives...
Ch. 6.5 - Prob. 6.11PCh. 6.5 - Prob. 6.12PCh. 6.5 - What alkene with the molecular formula C6H12, when...Ch. 6 - Prob. 6.15PCh. 6 - Prob. 6.16PCh. 6 - Predict the organic product(s) of the reaction of...Ch. 6 - Prob. 6.18PCh. 6 - Prob. 6.20PCh. 6 - Draw a structural formula for an alkene with the...Ch. 6 - Account for the fact that addition of HCl to...Ch. 6 - Account for the fact that treating propenoic acid...Ch. 6 - Draw a structural formula for the alkene with the...Ch. 6 - Draw the alternative chair conformations for the...Ch. 6 - Draw a structural formula for the cycloalkene with...Ch. 6 - Reaction of this bicycloalkene with bromine in...Ch. 6 - Terpin, prepared commercially by the...Ch. 6 - Propose a mechanism for this reaction and account...Ch. 6 - Treating 2-methylpropene with methanol in the...Ch. 6 - When 2-pentene is treated with Cl2 in methanol,...Ch. 6 - Treating cyclohexene with HBr in the presence of...Ch. 6 - Propose a mechanism for this reaction. 1-Pentane...Ch. 6 - Treating 4-penten-1-ol with bromine in water forms...Ch. 6 - Prob. 6.35PCh. 6 - Prob. 6.36PCh. 6 - Reaction of -pinene with borane followed by...Ch. 6 - Write structural formulas for the major organic...Ch. 6 - Draw the structural formula of the alkene that...Ch. 6 - Consider the following reaction. (a) Draw a...Ch. 6 - Prob. 6.42PCh. 6 - Prob. 6.43PCh. 6 - Show how to convert ethylene to these compounds....Ch. 6 - Show how to convert cyclopentene into these...Ch. 6 - Prob. 6.46PCh. 6 - Describe the stereochemistry of the bromohydrin...Ch. 6 - Prob. 6.49PCh. 6 - Treating 1,3-butadiene with 1 mole of HBr gives a...Ch. 6 - In this chapter, we studied the mechanism of the...Ch. 6 - As we have seen in this chapter, carbon-carbon...Ch. 6 - Prob. 6.53PCh. 6 - Prob. 6.54P
Knowledge Booster
Similar questions
- Following is a retrosynthetic scheme for the synthesis of the tricyclic diene on the left. Show how to accomplish this synthesis from 2-bromopropane, cyclopentadiene, and 2-cyclohexenone.arrow_forwardSuppose you were told that each reaction is a substitution reaction, but you were not told the mechanism. Describe how you could conclude from the structure of the haloalkane or cycloalkene, the nucleophile, and the solvent that each reaction is an SN1 reaction.arrow_forwardWe'll see in the next chapter that the stability of carbocations depends on the number of alkyl substituents attached to the positively charged carbon - the more alkyl substituents there are, the more stable the cation. Which of the two carbocations in each pair is more stable? If they are of equal stablility, specify this.arrow_forward
- In parts 1 and 2 draw the two organic products of this reaction, showing any nonzero formal charges. Then, in part 3 answer the question regarding purification of the reaction mixture. 1. Draw the product with the higher molecular weight here: 2. Draw the product with the lower molecular weight here:arrow_forwardSuppose you are told that each reaction is a substitution reaction but are not told the mechanism. Describe how you could conclude from the structure of the haloalkane, the nucleophile, and the solvent that each reaction is an SN2 reaction.arrow_forwardDraw structural formulas for the isomeric carbocation intermediates formed on treatment of each alkene with HCl. Label each carbocation 1°, 2°, or 3° and state which of the isomeric carbocations forms more readily.arrow_forward
- Show the process of synthesizing 2-methyl2-cyclohexenone from 2-methyl cyclohexanone. Write down the stages of the monocyclic reaction process in detail.arrow_forwardAre phenyl carbocations more stable than tertiary carbocations? Is there a carbocation that is more stable than a tertiary carbocation? If yes, what is the reason why it is more stablearrow_forwardWhen 1 mole of buta-1,3-diene reacts with 1 mole of HBr, both 3-bromobut-1-ene and 1-bromobut-2-ene are formed. Propose a mechanism to account for this mixture of products.arrow_forward
- Arrange the following alky halides in order of decreasing reactivity in an SN1 reaction: 2-bromopentane, 2-chloropentane, 1-chloropentane, 3-bromo-3-methylpentanearrow_forwardQuestion: How do quantum mechanical effects influence the stability and reactivity of molecules with non-classical carbocations, such as the 2-norbornyl cation, and how does this impact the reaction mechanisms and outcomes?arrow_forwardComplete these reactions, showing the stereochemistry of the productarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
