
Chemistry & Chemical Reactivity
9th Edition
ISBN: 9781133949640
Author: John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 6, Problem 72IL
The spectrum shown here is for aspirin. The vertical axis is the amount of light absorbed, and the horizontal axis is the wavelength of incident light (in nm). (For more on spectrophotometry, see Section 4.9)
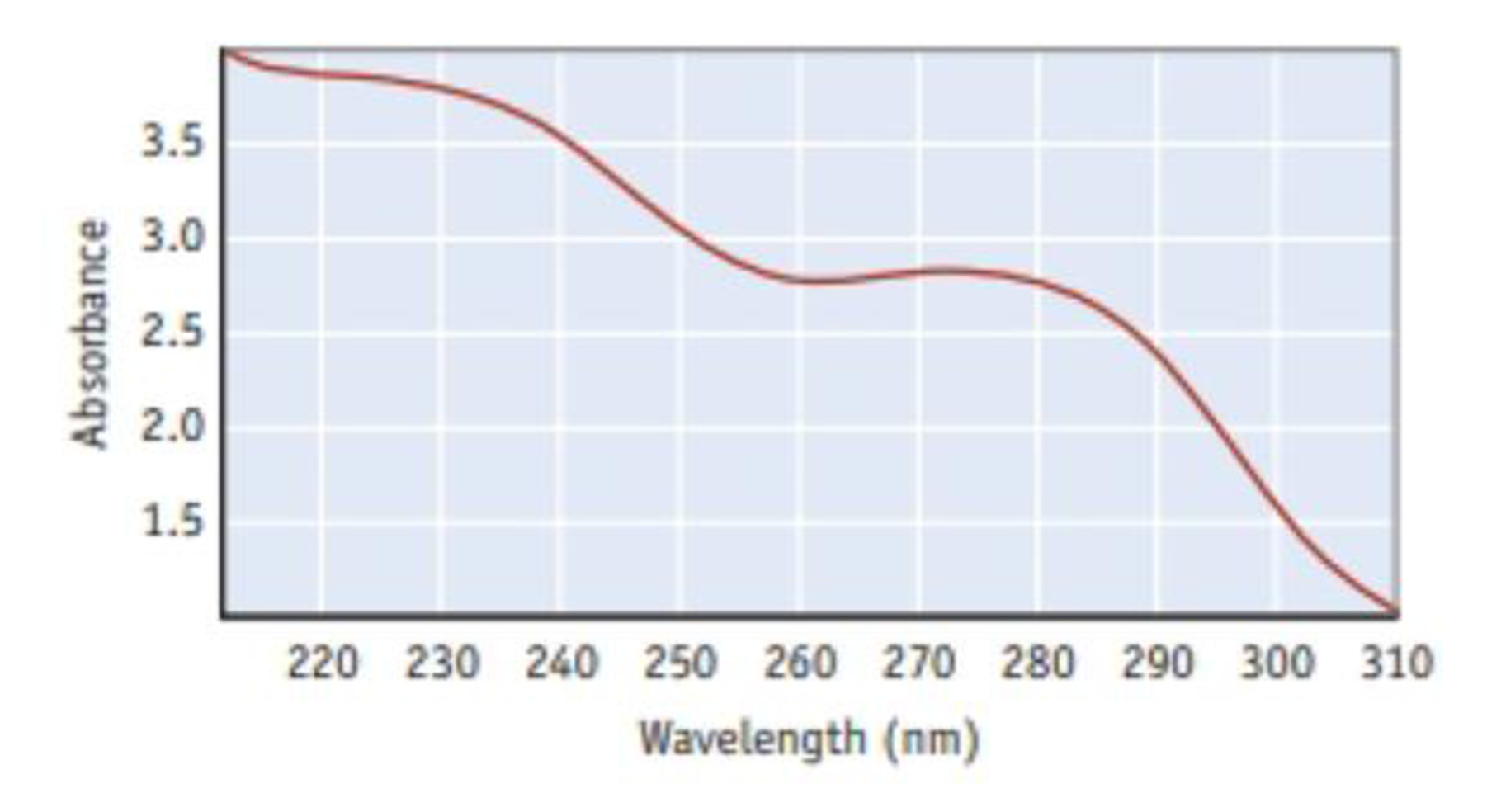
What is the frequency of
Expert Solution & Answer
Trending nowThis is a popular solution!

Chapter 6 Solutions
Chemistry & Chemical Reactivity
Ch. 6.1 - (a) Which color in the visible spectrum has the...Ch. 6.1 - 1. Which of the following types of electromagnetic...Ch. 6.1 - What is the wavelength of an FM radio signal that...Ch. 6.2 - Calculate the energy per mole of photons for the...Ch. 6.2 - Prob. 1RCCh. 6.2 - Prob. 2RCCh. 6.3 - Prob. 1CYUCh. 6.3 - The Lyman series of spectral lines for the H atom,...Ch. 6.3 - 1. Based on Bohr's theory, which of the following...Ch. 6.3 - Based on Bohr's theory, which of the following...
Ch. 6.4 - Calculate the wavelength associated with a neutron...Ch. 6.4 - 1. The wavelength associated with an electron...Ch. 6.5 - 1. What label is given to an orbital with quantum...Ch. 6.5 - 2. How many orbitals are in the n = 4...Ch. 6.5 - Prob. 3RCCh. 6.6 - 1. Which of the following is not a correct...Ch. 6.6 - Which of the following sets of quantum numbers...Ch. 6.6 - How many nodal planes exist for a 5d orbital? (a)...Ch. 6.7 - Which of the following is a valid set of quantum...Ch. 6 - Answer the following questions based on Figure...Ch. 6 - Consider the colors of the visible spectrum. (a)...Ch. 6 - Traffic signals are often now made of LEDs...Ch. 6 - Suppose you are standing 225 m from a radio...Ch. 6 - Green light has a wavelength of 5.0 102 nm. What...Ch. 6 - Violet light has wavelength of about 410 nm. What...Ch. 6 - The most prominent line in the emission spectrum...Ch. 6 - The most prominent line in the emission spectrum...Ch. 6 - Place the following types of radiation in order of...Ch. 6 - Place the following types of radiation in order of...Ch. 6 - An energy of 3.3 1019 J/atom is required to cause...Ch. 6 - You are an engineer designing a switch that works...Ch. 6 - The most prominent line in the spectrum of mercury...Ch. 6 - The most prominent line in the spectrum of neon is...Ch. 6 - A line in the Balmer series of emission lines of...Ch. 6 - What are the wavelength and frequency of the...Ch. 6 - Consider only transitions involving the n = 1...Ch. 6 - Consider only transitions involving the n = 1...Ch. 6 - The energy emitted when an electron moves from a...Ch. 6 - If energy is absorbed by a hydrogen atom in its...Ch. 6 - Calculate the wavelength and frequency of light...Ch. 6 - Calculate the wavelength and frequency of light...Ch. 6 - An electron moves with a velocity of 2.5 X 108...Ch. 6 - A beam of electrons (m = 9.11 X 1031 kg/electron)...Ch. 6 - Calculate the wavelength, in nanometers,...Ch. 6 - A rifle bullet (mass = 1.50 g) has a velocity of...Ch. 6 - (a) When n = 4, what are the possible values of ?...Ch. 6 - (a) When n = 4, = 2, and m = 1, to what orbital...Ch. 6 - A possible excited state of the H atom has the...Ch. 6 - A possible excited state for the H atom has an...Ch. 6 - How many subshells occur in the electron shell...Ch. 6 - Prob. 32PSCh. 6 - Explain briefly why each of the following is not a...Ch. 6 - Which of the following represent valid sets of...Ch. 6 - What is the maximum number of orbitals that can be...Ch. 6 - What is the maximum number of orbitals that can be...Ch. 6 - Explain briefly why each of the following is not a...Ch. 6 - Explain briefly why each of the following is not a...Ch. 6 - State which of the following orbitals cannot exist...Ch. 6 - State which of the following orbitals cannot exist...Ch. 6 - Write a complete set of quantum numbers (n, , m)...Ch. 6 - Write a complete set of quantum numbers (n, , and...Ch. 6 - A particular orbital has n = 4 and = 2. What must...Ch. 6 - A given orbital has a magnetic quantum number of m...Ch. 6 - Prob. 45PSCh. 6 - Prob. 46PSCh. 6 - Which of the following are applicable when...Ch. 6 - Prob. 48GQCh. 6 - Give the number of nodal surfaces through the...Ch. 6 - What is the maximum number of s orbitals found in...Ch. 6 - Match the values of l shown in the table with...Ch. 6 - Sketch a picture of the 90% boundary surface of an...Ch. 6 - Complete the following table.Ch. 6 - Excited H atoms have many emission lines. One...Ch. 6 - An advertising sign gives off red light and green...Ch. 6 - Radiation in the ultraviolet region of the...Ch. 6 - A cell phone sends signals at about 850 MHz (where...Ch. 6 - Assume your eyes receive a signal consisting of...Ch. 6 - If sufficient energy is absorbed by an atom, an...Ch. 6 - Suppose hydrogen atoms absorb energy so that...Ch. 6 - Rank the following orbitals in the H atom in order...Ch. 6 - How many orbitals correspond to each of the...Ch. 6 - Cobalt-60 is a radioactive isotope used in...Ch. 6 - Exposure to high doses of microwaves can cause...Ch. 6 - When the Sojourner spacecraft landed on Mars in...Ch. 6 - The most prominent line in the emission spectrum...Ch. 6 - Answer the following questions as a summary quiz...Ch. 6 - Answer the following questions as a summary quiz...Ch. 6 - For an electron in a hydrogen atom, calculate the...Ch. 6 - A solution of KMnO4 absorbs light at 540 nm (page...Ch. 6 - Prob. 71ILCh. 6 - The spectrum shown here is for aspirin. The...Ch. 6 - The infrared spectrum for methanol. CH3OH, is...Ch. 6 - Bohr pictured the electrons of the atom as being...Ch. 6 - Light is given off by a sodium- or...Ch. 6 - Prob. 76SCQCh. 6 - What does wave-particle duality mean? What are its...Ch. 6 - Prob. 79SCQCh. 6 - Suppose you live in a different universe where a...Ch. 6 - A photon with a wavelength of 93.8 nm strikes a...Ch. 6 - Explain why you could or could not measure the...Ch. 6 - Prob. 83SCQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The eyes of certain reptiles pass a single visual signal to the brain when the visual receptors are struck by photons of a wavelength of 850 nm. If a total energy of 3.151014 J is required to trip the signal, what is the minimum number of photons that must strike the receptor?arrow_forwardConsider the colors of the visible spectrum. (a) Which colors of light involve less energy than green light? (b) Which color of light has photons of greater energy, yellow or blue? (c) Which color of light has the higher frequency, blue or green?arrow_forwardTraffic signals are often now made of LEDs (light-emitting diodes). Amber and green ones are pictured here. (a) The light from an amber signal has a wave-length of 595 nm, and that from a green signal has a wavelength of 500 nm. Which has the higher frequency? (b) Calculate the frequency of amber light.arrow_forward
- An FM radio station found at 103.1 on the FM dial broadcasts at a frequency of 1.031188s1 (103.1 MHz). What is the wavelength of these radio waves in meters?arrow_forwardThe most prominent line in the spectrum of mercury is at 253.652 nm. Other lines are located at 365.015 nm, 404.656 nm, 435.833 nm, and 101.975 nm. (a) which of these lines represents the most energetic light? (b) what is the frequency of the most prominent line? What is the energy of one photon with this wavelength? (c) Are any of these lines found in the spectrum of mercury shown in figure 6.6? What color or colors are these lines?arrow_forwardHuman color vision is " produced" by the nervous system based on how three different cone receptors interact with photons of light in the eye. These three different types of cones interact with photons of different frequency light, as indicated in the following chart: Cone Type Range of Light Frequency Detected S 6.00-7.49 1014s1 M 4.76-6.62 1014s1 L 4.28-6.00 1014s1 What wavelength ranges (and corresponding colors) do the three types of cones detect?arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStaxChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781337399074
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry: Principles and Practice
Chemistry
ISBN:9780534420123
Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward Mercer
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry: Matter and Change
Chemistry
ISBN:9780078746376
Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl Wistrom
Publisher:Glencoe/McGraw-Hill School Pub Co
Quantum Mechanics - Part 1: Crash Course Physics #43; Author: CrashCourse;https://www.youtube.com/watch?v=7kb1VT0J3DE;License: Standard YouTube License, CC-BY