
Concept explainers
(a)
Interpretation:
The formal charges of element A if it was in (a) Group 7A, (b) Group 5A, (c) Group 3A should be drawn.
Concept Introduction
- A formal charge (FC) is the charge assigned to an atom in a molecule, irrespective of relative electronegativity by thinking that electrons in all
chemical bonds are shared equally among atoms. - This method is used to identify the most probable Lewis structures if more than one possibility exists for a compound.
- Formal charge of an atom can be determined by the given formula.
To determine: Formal charge of the given species
(a)
Answer to Problem 5PPC
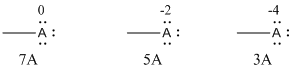
Explanation of Solution
Given Lewis structure
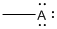
A formal charge (FC) is the charge assigned to an atom in a molecule, irrespective of relative electronegativity.
The formal charge of the given species is calculated,
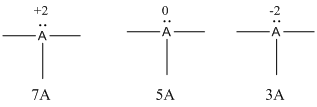
- If A is group 7A, then number of valence electrons is 7
Substituting these values to the equation,
- If A is group 5A, then number of valence electrons is 5
Substituting these values to the equation,
- If A is group 3A, then number of valence electrons is 3
Substituting these values to the equation,
Therefore,
The formal charges are,
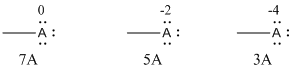
(b)
Interpretation:
The formal charges of element A if it was in (a) Group 7A, (b) Group 5A, (c) Group 3A should be drawn.
Concept Introduction
- A formal charge (FC) is the charge assigned to an atom in a molecule, irrespective of relative electronegativity by thinking that electrons in all chemical bonds are shared equally among atoms.
- This method is used to identify the most probable Lewis structures if more than one possibility exists for a compound.
- Formal charge of an atom can be determined by the given formula.
To determine: Formal charge of the given species
(b)
Answer to Problem 5PPC
Explanation of Solution
Given Lewis structure
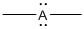
A formal charge (FC) is the charge assigned to an atom in a molecule, irrespective of relative electronegativity.
The formal charge of the given species is calculated,
- If A is group 7A, then number of valence electrons is 7
Substituting these values to the equation,
- If A is group 5A, then number of valence electrons is 5
Substituting these values to the equation,
- If A is group 3A, then number of valence electrons is 3
Substituting these values to the equation,
Therefore,
The formal charges are,

(c)
Interpretation:
The formal charges of element A if it was in (a) Group 7A, (b) Group 5A, (c) Group 3A should be drawn.
Concept Introduction
- A formal charge (FC) is the charge assigned to an atom in a molecule, irrespective of relative electronegativity by thinking that electrons in all chemical bonds are shared equally among atoms.
- This method is used to identify the most probable Lewis structures if more than one possibility exists for a compound.
- Formal charge of an atom can be determined by the given formula.
To determine: Formal charge of the given species
(c)
Answer to Problem 5PPC
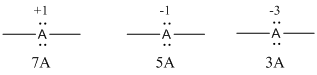
Explanation of Solution
Given Lewis structure
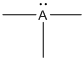
A formal charge (FC) is the charge assigned to an atom in a molecule, irrespective of relative electronegativity.
The formal charge of the given species is calculated,
- If A is group 7A, then number of valence electrons is 7
Substituting these values to the equation,
- If A is group 5A, then number of valence electrons is 5
Substituting these values to the equation,
- If A is group 3A, then number of valence electrons is 3
Substituting these values to the equation,
Therefore,
The formal charges are,

Want to see more full solutions like this?
Chapter 6 Solutions
Chemistry: Atoms First
- Based on the concept of formal charge, what is the central atom in (a) HCN (do not include H as a possibility)? b) NOCI (Cl is always a terminal atom)?arrow_forwardWhat is the formal charge on nitrogen in the anionic molecule (NO2)-?arrow_forwardHow many covalent bonds are there in ClO2? Assume Cl will expand its octet to minimize formal charges.arrow_forward
- In the Lewis structure shown here, A, D, E, Q, X, and Z representelements in the first two rows of the periodic table.Identify all six elements so that the formal charges of all atomsare zero.arrow_forwardHow many resonance structures can be drawn for the hydrogen tellurate ion (HTeO4–) in which the central tellurium atom bears a –1 formal charge and the oxygens bear formal charges of either zero or –1? Enter your answer as a whole number.arrow_forwardWhat is the formal charge of every atom in Cl2?arrow_forward
- Calculate the formal charge of all the atoms in CO, CO2 and CO32- (you will need to draw the correct Lewis structure first)arrow_forwardWhat is the formal charge on carbon in COCl2?arrow_forwardFor the carbonate ion (CO32-), there are 3 resonance structures. For any given structure what would the formal charge be for an oxygen that has a single bond to the central carbon atom?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning


