
Interpretation:
Given problem “How many molecules of
Concept Introduction:
Using the

Molar mass of a substance gives a relationship between the number of grams and number of moles of that particular substance.

The Chemical formula subscript also provides relationship between the number of moles of substance to the number of moles of its individual components present in the substance.

All the three above relationship can be put into a single diagram as shown below,
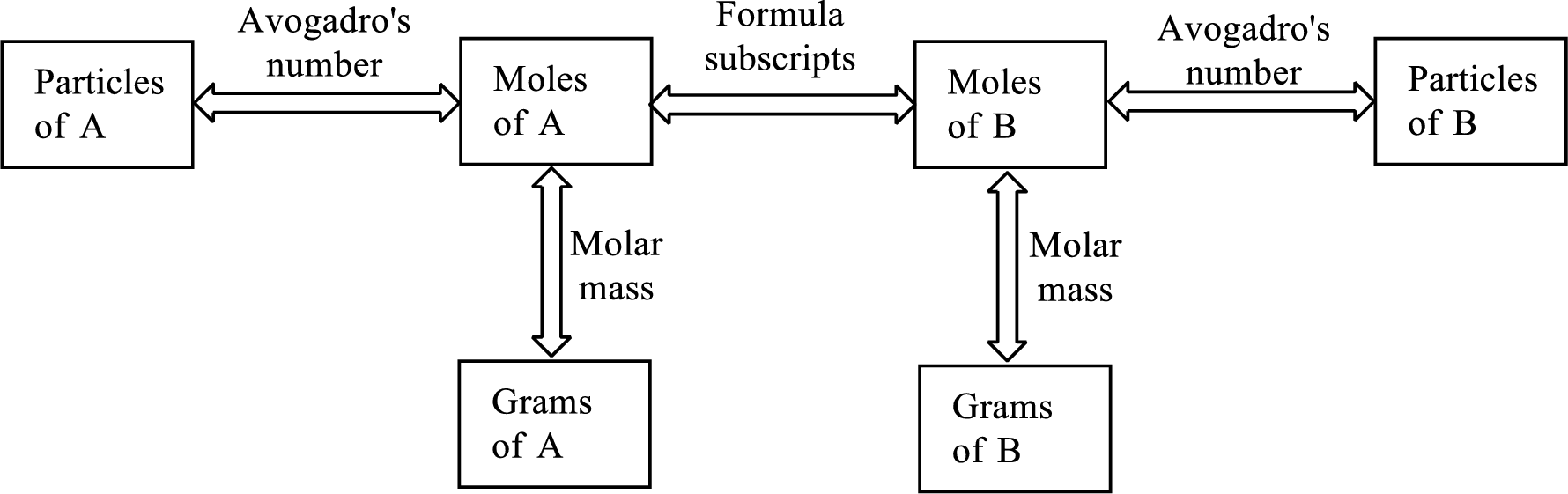
Want to see the full answer?
Check out a sample textbook solution
Chapter 6 Solutions
General, Organic, and Biological Chemistry
- topic: avogadro's number, mole, molar mass benadryl (chemical formula C17H21NO) show calculations for the following a.) how many moles of carbon are present in 0.733 mole of benadryl? b.) how many moles of hydrogen are present in 35.6 grams of benadryl? c.) how many atoms of nitrogren are present in 25.0 mg of benadryl? d.) how many molecules of benadryl are present in 1250 mcg of benadryl?arrow_forwardAnswer the following questions on Avogadro's Number and the mole. (SHOW WORK and round ALL final answers to 2 decimal places) How many molecules are in 0.25 moles of CH4? How many total atoms are in 1.0 moles of H2O?arrow_forward2. Calculate the number of moles and grams from the following: (use correct sig figs in answer) a. 3.510 x 1022 molecules of PCl5 b. 4.386 x 1023 molecules of MgSO4arrow_forward
- 1. Find the number of moles and molecules from the following: Avogadro's number is 6.022 x 1023 molecules / mole (Use correct sig figs in answer) a. 86.5g Al(ClO3)3 b. 50.0g BaCl2arrow_forwardMole problem (Chemistry), Please show all calculations: How many moles of carbon tetrafluoride are there in a 1.55 * 105 kg sample of carbon tetrafluoride?arrow_forwardHow many grams of Cr2(SO4)3) will be produced from 25.61g of K2Cr2O7 ? Request Only correct and typed answer pleasearrow_forward
- What is the mass of 1.76 mol of BCl3? (Answer in grams)arrow_forwardPlease help!! 1) 42 g of OCl2 2) 4.30 moles of CCl4 3) 3.76 × 10^34 molecules of SeCl4. Calculate the no. Of chlorine atom in each of the sample.arrow_forwardow many MOLES of ammonium chromate are present in 2.14 grams of this compound ? moles. Please type answer note write by hendarrow_forward
- Oxygen was discovered by Joseph Priestley in 1774 when he heated mercury(II) oxide to decompose it to form its constituent elements. How much mercury(II) oxide (HgO) is needed to produce 492 g of oxygen (O2)? Answer in units of mol. How much mercury is produced? Answer in units of mol.arrow_forwardHow many atoms of magnesium mg in 2.4g of magnesium ? Show steparrow_forwardcalculate the mass in grams of 14.53 mol of carbon tetrachloride (CCl4) answer in scientific notationarrow_forward
- Chemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
