
Chemistry: An Atoms First Approach
2nd Edition
ISBN: 9781305079243
Author: Steven S. Zumdahl, Susan A. Zumdahl
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 7, Problem 113AE
Compare your answer from Exercise 72 of Chapter 3 to the ∆H value calculated from standard enthalpies of formation in Appendix 4. Explain any discrepancies.
…..
72. Acetic acid is responsible for the sour taste of vinegar. It can be manufactured using the following reaction:
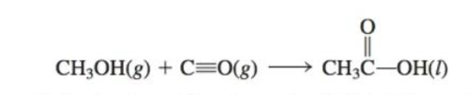
Use tabulated values of bond engines(Table 3-3) to estimate ∆E for this reaction
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Using the table of bond enthalpies in your textbook, predict ΔrH for the following reaction. Express your answer in kJ/mol.
HCΞCH + 2 H2 → H3C-CH3
From the following enthalpies of reaction:
H2(g)+F2(g)→2HF(g),ΔH=−537kJ
C(s)+2F2(g)→CF4(g),ΔH=−680kJ
2C(s)+2H2(g)→C2H4(g),ΔH=+52.3kJ
calculate ΔH for the reaction of ethylene with F2:
C2H4(g)+6F2(g)→2CF4(g)+4HF(g)
If hydrogen were used as a fuel, it could be burned according to this reaction:
H2(g) + 1/2 O2(g) --------> H2O(g)
Use average bond energies to calculate ∆Hrxn for this reaction and also for the combustion of methane (CH4). Which fuel yields more energy per mole? Per gram?
Chapter 7 Solutions
Chemistry: An Atoms First Approach
Ch. 7 - Define the following terms: potential energy,...Ch. 7 - Consider the following potential energy diagrams...Ch. 7 - What is the first law of thermodynamics? How can a...Ch. 7 - When a gas expands, what is the sign of w? Why?...Ch. 7 - Prob. 5RQCh. 7 - High-quality audio amplifiers generate large...Ch. 7 - Explain how calorimetry works to calculate H or E...Ch. 7 - What is Hesss law? When a reaction is reversed,...Ch. 7 - Define the standard enthalpy of formation. What...Ch. 7 - Prob. 1ALQ
Ch. 7 - Prob. 2ALQCh. 7 - A fire is started in a fireplace by striking a...Ch. 7 - Liquid water turns to ice. Is this process...Ch. 7 - Prob. 5ALQCh. 7 - Prob. 6ALQCh. 7 - Consider 5.5 L of a gas at a pressure of 3.0 atm...Ch. 7 - Explain why oceanfront areas generally have...Ch. 7 - Hesss law is really just another statement of the...Ch. 7 - Prob. 10ALQCh. 7 - Prob. 11QCh. 7 - Prob. 12QCh. 7 - Assuming gasoline is pure C8H18(l), predict the...Ch. 7 - Prob. 14QCh. 7 - The enthalpy change for the reaction...Ch. 7 - For the reaction HgO(s)Hg(l)+12O2(g),H=+90.7KJ: a....Ch. 7 - Prob. 17QCh. 7 - The enthalpy change for a reaction is a state...Ch. 7 - Standard enthalpies of formation are relative...Ch. 7 - The combustion of methane can be represented as...Ch. 7 - Prob. 21QCh. 7 - Prob. 22QCh. 7 - Prob. 23QCh. 7 - Prob. 24QCh. 7 - Prob. 25ECh. 7 - Prob. 26ECh. 7 - Consider the following diagram when answering the...Ch. 7 - Consider the accompanying diagram. Ball A is...Ch. 7 - A gas absorbs 45 kJ of heat and does 29 kJ of...Ch. 7 - A system releases 125 kJ of heat while 104 kJ of...Ch. 7 - Calculate E for each of the following. a. q = 47...Ch. 7 - A system undergoes a process consisting of the...Ch. 7 - If the internal energy of a thermodynamic system...Ch. 7 - Calculate the internal energy change for each of...Ch. 7 - A sample of an ideal gas at 15.0 atm and 10.0 L is...Ch. 7 - Prob. 36ECh. 7 - Consider a mixture of air and gasoline vapor in a...Ch. 7 - As a system increases in volume, it absorbs 52.5 J...Ch. 7 - A balloon filled with 39.1 moles of helium has a...Ch. 7 - Prob. 40ECh. 7 - One of the components of polluted air is NO. It is...Ch. 7 - Prob. 42ECh. 7 - Are the following processes exothermic or...Ch. 7 - Are the following processes exothermic or...Ch. 7 - The overall reaction in a commercial heat pack can...Ch. 7 - Consider the following reaction:...Ch. 7 - Consider the combustion of propane:...Ch. 7 - Consider the following reaction:...Ch. 7 - Prob. 49ECh. 7 - The specific heat capacity of silver is 0.24 J/Cg....Ch. 7 - A 500-g sample of one of the substances listed in...Ch. 7 - Prob. 52ECh. 7 - A 30.0-g sample of water at 280. K is mixed with...Ch. 7 - A biology experiment requires the preparation of a...Ch. 7 - A 5.00-g sample of aluminum pellets (specific heat...Ch. 7 - Hydrogen gives off 120. J/g of energy when burned...Ch. 7 - Prob. 57ECh. 7 - A 110.-g sample of copper (specific heat capacity...Ch. 7 - In a coffee-cup calorimeter, 50.0 mL of 0.100 M...Ch. 7 - In a coffee-cup calorimeter, 100.0 mL of 1.0 M...Ch. 7 - A coffee-cup calorimeter initially contains 125 g...Ch. 7 - In a coffee-cup calorimeter, 1.60 g NH4NO3 is...Ch. 7 - Consider the dissolution of CaCl2:...Ch. 7 - Consider the reaction...Ch. 7 - The heat capacity of a bomb calorimeter was...Ch. 7 - The combustion of 0.1584 g benzoic acid increases...Ch. 7 - The enthalpy of combustion of solid carbon to form...Ch. 7 - Combustion reactions involve reacting a substance...Ch. 7 - Given the following data calculate H for the...Ch. 7 - Given the following data...Ch. 7 - Prob. 71ECh. 7 - Calculate H for the reaction...Ch. 7 - Given the following data...Ch. 7 - Given the following data...Ch. 7 - Give the definition of the standard enthalpy of...Ch. 7 - Write reactions for which the enthalpy change will...Ch. 7 - Prob. 77ECh. 7 - Use the values of Hf in Appendix 4 to calculate H...Ch. 7 - The Ostwald process for the commercial production...Ch. 7 - Calculate H for each of the following reactions...Ch. 7 - The reusable booster rockets of the space shuttle...Ch. 7 - The space shuttle Orbiter utilizes the oxidation...Ch. 7 - Consider the reaction...Ch. 7 - The standard enthalpy of combustion of ethene gas,...Ch. 7 - Water gas is produced from the reaction of steam...Ch. 7 - Prob. 86ECh. 7 - Prob. 87ECh. 7 - Prob. 88ECh. 7 - Some automobiles and buses have been equipped to...Ch. 7 - The complete combustion of acetylene, C2H2(g),...Ch. 7 - Prob. 91AECh. 7 - One way to lose weight is to exercise! Walking...Ch. 7 - Three gas-phase reactions were run in a...Ch. 7 - Nitrogen gas reacts with hydrogen gas to form...Ch. 7 - Combustion of table sugar produces CO2(g) and H2O(...Ch. 7 - Prob. 96AECh. 7 - Consider the following cyclic process carried out...Ch. 7 - Calculate H for the reaction...Ch. 7 - The enthalpy of neutralization for the reaction of...Ch. 7 - Prob. 100AECh. 7 - If a student performs an endothermic reaction in a...Ch. 7 - In a bomb calorimeter, the reaction vessel is...Ch. 7 - The bomb calorimeter in Exercise 102 is filled...Ch. 7 - Prob. 104AECh. 7 - Consider the following equations:...Ch. 7 - Prob. 106AECh. 7 - At 298 K, the standard enthalpies of formation for...Ch. 7 - Prob. 108AECh. 7 - A sample of nickel is heated to 99.8C and placed...Ch. 7 - Quinone is an important type of molecule that is...Ch. 7 - Calculate H for each of the following reactions,...Ch. 7 - Compare your answers from parts a and b of...Ch. 7 - Compare your answer from Exercise 72 of Chapter 3...Ch. 7 - Consider a balloon filled with helium at the...Ch. 7 - Prob. 115CWPCh. 7 - Prob. 116CWPCh. 7 - Prob. 117CWPCh. 7 - A swimming pool, 10.0 m by 4.0 m, is filled with...Ch. 7 - Prob. 119CWPCh. 7 - Calculate H for the reaction...Ch. 7 - Which of the following substances have an enthalpy...Ch. 7 - Consider 2.00 moles of an ideal gas that are taken...Ch. 7 - For the process H2O(l)H2O(g) at 298 K and 1.0 atm,...Ch. 7 - The sun supplies energy at a rate of about 1.0...Ch. 7 - Prob. 125CPCh. 7 - The standard enthalpies of formation for S(g),...Ch. 7 - Use the following standard enthalpies of formation...Ch. 7 - The standard enthalpy of formation for N2H4(g) is...Ch. 7 - The standard enthalpy of formation for NO(g) is...Ch. 7 - A piece of chocolate cake contains about 400...Ch. 7 - You have a l.00-mole sample of water at 30.C and...Ch. 7 - A 500.0-g sample of an element at 195C is dropped...Ch. 7 - A cubic piece of uranium metal (specific heat...Ch. 7 - On Easter Sunday, April 3, 1983, nitric acid...Ch. 7 - Using data from Chapter 2, calculate the change in...Ch. 7 - In Exercise 89 in Chapter 3, the Lewis structures...Ch. 7 - A gaseous hydrocarbon reacts completely with...
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- The bond energy for a CH bond is about 413 kJ/mol in CH4 but 380 kJ/mol in CHBr3. Although these values are relatively close in magnitude, they are different. Explain why they are different. Does the fact that the bond energy is lower in CHBr3, make any sense? Why?arrow_forwardAt 298 K, the standard enthalpies of formation for C2H2(g) and C6H6(l) are 227 kJ/mol and 49 kJ/mol, respectively. a. Calculate H for C6H6(l)3C2H2(g) b. Both acetylene (C2H2) and benzene (C6H6) can be used as fuels. Which compound would liberate more energy per gram when combusted in air?arrow_forwardGiven the following data: NO2(g) NO(g) + O(g)H = 233 kJ 2O3(g) 3O2(g)H = 427 kJ NO(g) + O3(g) NO2(g) + O2(g)H = 199 kJ Calculate the bond energy for the O2 bond, that is, calculate H for: O2(g) 2O(g)H = ?arrow_forward
- Would the amount of heat absorbed by the dissolution in Example 5.6 appear greater, lesser, or remain the same if the heat capacity of the calorimeter were taken into account? Explain your answer.arrow_forwardThe standard enthalpies of formation for S(g), F(g), SF4(g), and SF6(g) are +278.8, +79.0, 775, and +1209 KJ/mol, respectively. a. Use these data to estimate the energy of an SF bond. b. Compare your calculated value to the value given in Table 3-3. What conclusions can you draw? c. Why are the Hf values for S(g) and F(g) not equal to zero, since sulfur and fluorine are elements?arrow_forwardUse the molar bond enthalpy data in the table to estimate the value of Δ?∘rxnΔHrxn° for the equation C2H4(g)+HBr(g)⟶C2H5Br(g) The bonding in the molecules is shown. Average molar bond enthalpies. (?bond)(Hbond) Bond ??⋅???−1kJ⋅mol−1 Bond ??⋅???−1kJ⋅mol−1 O−H 464 C≡N 890 O−O 142 N−H 390 C−O 351 N−N 159 O=O 502 N=N 418 C=O 730 N≡N 945 C−C 347 F−F 155 C=C 615 Cl−Cl 243 C≡C 811 Br−Br 192 C−H 414 H−H 435 C−F 439 H−F 565 C−Cl 331 H−Cl 431 C−Br 276276 H−Br 368 C−N 293293 H−S 364 C=N 615615 S−S 225 Δ?∘rxn= kJarrow_forward
- Use the data found in Thermodynamic Properties and the data in Bond Energies. to estimate the enthalpies of formation of the following substances: (Hint: the standard state of carbon is not a gas.) Please see https://youtu.be/d3H2Qnmp0BQ for assistance. (a) NF3(g) kJ(b) H2N-NH2(g) kJ(c) F2CCH2(g) kJ(d) CH3Cl(g) kJarrow_forwardGiven that Δ?∘f[H(g)]=218.0 kJ⋅mol−1 Δ?∘f[C(g)]=716.7 kJ⋅mol−1 Δ?∘f[CH4(g)]=−74.6 kJ⋅mol−1 calculate the average molar bond enthalpy of the carbon–hydrogen bond in a CH4CH4 molecule. Δ?C−H=ΔHC−H= kJ⋅mol−1arrow_forwardCl₂ is a stable diatomic molecule. It can be decomposed to form two Cl atoms as shown below. Cl₂ → 2Cl(g) Predict the change in free energy (∆G) of this reaction.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:9781305079243
Author:Steven S. Zumdahl, Susan A. Zumdahl
Publisher:Cengage Learning


Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:9781938168390
Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:OpenStax

Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:9781133949640
Author:John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:Cengage Learning
Calorimetry Concept, Examples and Thermochemistry | How to Pass Chemistry; Author: Melissa Maribel;https://www.youtube.com/watch?v=nSh29lUGj00;License: Standard YouTube License, CC-BY