
Concept explainers
Interpretation:Whether in the model of 1,2-dimethylcyclopentane, the molecule in the left box is same as the molecule in the right box or not needs to be determined.
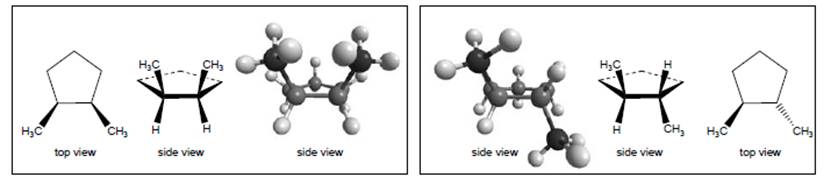
Concept Introduction:
The cis and trans isomerism concept will be applied here. Any molecule will be called cis if it follows the conditions for itthat is the same groups attached either above the plane or below the plane.For trans isomer,same groups are attached where one is above the plane and other group is below the plane or vice versa.
Answer to Problem 1CTQ
The given both structures of isomers of 1,2 dimethylcyclopentane are similar to each other. This is because when the bonds of the groups interchanges with each other, the bond’s spatial character changes. As a result of which these structures can be converted to each other without breaking of bonds.
Explanation of Solution
It is given that the left box and right box contain two molecules of 1,2-dimethylcyclopentane.Thus, it is very easy to conclude that the 2 different molecules are isomers of 1,2-dimethylcyclopentane. This is because both the molecules have same number of atoms in it.
Both the molecules are similar toeach other. Thiscan be explained as, if the interchanging of bonds takes place, similar structure can be obtained. In the interchanging of structure in left box and right box, there is no breaking of bonds takes place. For example, if the methyl group attached to the bond below the planeis interchanged with other group attached to the same atom, then the position of methyl group will be converted to above the plane from below the plane. In this process, no breaking of bond takes place then similar structure will be obtained.
Thus, the structure on the left box is similar to that on the right box.
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry: A Guided Inquiry
- Without counting hydrogens, determine which one of the following CANNOT be the unknownmolecule with molecular formula C7H8NOBr , and explain your reasoning.arrow_forwardDraw the most stable conformation of the molecule while taking into account the approximate values of the energies of the following skew interactions:arrow_forwardas far as i know all the chiral molecule can be used to carry out optical resolution. so i think material C also can be an option but the answer says only E can be used. could you explain why not C? Please answer very soon will give rating surelyarrow_forward
- In the previous problem, you saw that there were two chiral carbons in 4-bromo-3-hydroxypentanal. That molecule is drawn below with the chiral carbons circled. What is the maximum number of stereoisomers that can exist for this molecule? Can you help me, please? Can you explain to me?arrow_forwardImage below, Why is this not a constructual isomer?arrow_forwardOn b), I understand that we don't know which side of the molecule the Cl would attach on, but I was under the impression that we only show stereochemistry on a chiral center, and I do not see where the chiral center is on this molecule. Would you be able to explain this?arrow_forward
- Assign the stereochemical configuration (R or S) for each molecule. For this question, the priorities have already been assigned.arrow_forwardCan you give me detail about the chiral carbon step by step with an examples, please?arrow_forwardDraw an exact 3-D representation of the molecule from the model. please answer all question step by step. Answer follow image: Please must mention: valency electron, molecular geometry handwritten answerarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
