
Concept explainers
Consider the following
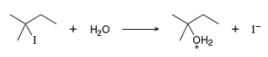
a. Draw a mechanism for this reaction using curved arrows.
b. Draw an energy diagram. Label the axes, the reactants, products,
c. Draw the structure of any transition states.
d. What is the rate equation for this reaction?
e. What happens to the reaction rate in each of the following instances? [1] The leaving group is changed from
(a)
Interpretation: The mechanism of the given reaction is to be drawn by the use of curved arrows.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron rich chemical species that contains negative charge or lone pair of electrons are known as a nucleophile. In a nucleophilic substitution reaction, nucleophile takes the position of leaving group by attacking the electron deficient carbon atom.
Answer to Problem 7.58P
The mechanism of the given reaction is shown in Figure 1.
Explanation of Solution
The structure of the given alkyl halide shows that carbon atom, on which bromine is present, is bonded to three other carbon atoms. Hence, the bromine atom is bonded to tertiary carbon atom and the given alkyl halide is
In
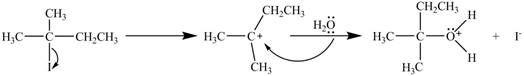
Figure 1
The mechanism of the given reaction is shown in Figure 1.
(b)
Interpretation: The energy diagram is to be drawn. The axes, reactants, products,
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The graphical representation of chemical reaction in which x-axis represents energy of the reaction and y-axis represents the reaction coordinate is called energy profile diagram.
Answer to Problem 7.58P
The energy diagram of the given reaction is shown in Figure 2.
Explanation of Solution
In
The energy of products is equal to the energy of reactants. Therefore, the energy diagram of the given reaction is,
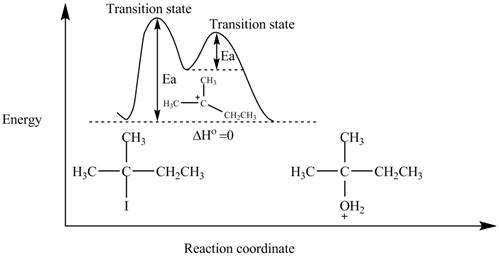
Figure 2
The x-axes of the energy diagram represent the energy of reactants and products and y-axes represent the reaction coordinate.
The energy diagram of the given reaction is shown in Figure 2.
(c)
Interpretation: The structure of the transition state is to be drawn.
Concept introduction: In
Answer to Problem 7.58P
The structure of the transition states is shown in Figure 3.
Explanation of Solution
In
Therefore, the transition states of the given reaction are,
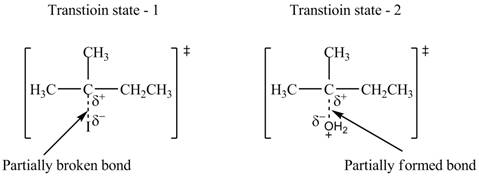
Figure 3
The structure of the transition states is shown in Figure 3.
(d)
Interpretation: The rate equation of the given reaction is to be predicted.
Concept introduction: The rate of
The rate of
Answer to Problem 7.58P
The rate equation of the given reaction is,
Explanation of Solution
The rate of
The rate of
The alkyl halide of given reaction is
Therefore, the rate equation of given reaction is,
The rate equation of the given reaction is,
(e)
Interpretation: The change that occurs to the reaction rate in given instances is to be stated.
Concept introduction: The rate of
The rate of
Answer to Problem 7.58P
The change that occurs to the reaction rate in given instances is,
[1] The rate of the reaction will decrease.
[2] The rate of the reaction will decrease.
[3] The rate of the reaction will decrease.
[4] The rate of the reaction remains unchanged.
[5] The rate of the reaction increases by five times.
Explanation of Solution
[1]
The tertiary halide undergoes nucleophilic substitution by
[2]
The polar protic solvent favors
[3]
The tertiary halide undergoes nucleophilic substitution by
[4]
The rate of
The rate of
According the given statement, the concentration of
Therefore, the rate of the reaction remains unchanged when the concentration of
[5]
The rate of
The rate of
According the given statement, the concentration of
Therefore, the rate of the reaction increases by five times when the concentration
The change that occurs to the reaction rate in given instances is,
[1] The rate of the reaction will decrease.
[2] The rate of the reaction will decrease.
[3] The rate of the reaction will decrease.
[4] The rate of the reaction remains unchanged.
[5] The rate of the reaction increases by five times.
Want to see more full solutions like this?
Chapter 7 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Inorganic Chemistry
General, Organic, and Biological Chemistry - 4th edition
Chemistry
Basic Chemistry
General Chemistry: Atoms First
- The Br produced by the reaction in Figure 9.2 could combine with the carbocation you drew to give a dibromide product (a product with two Br atoms). Use a curved arrow to show the Br making a bond to the carbocation, and draw the resulting dibromide product.arrow_forwardThese synthesis reactions can be carried out in three or fewer steps. Fill in the reagents required for each step. If a step is not needed, write an X for that step.arrow_forwardDraw the rearrangement mechanism for the following image #10arrow_forward
- Please choose which product would result from the reaction.True or False?: Using Cl2/hν in place of Br2/hν on the same compound seen above would yield only one product.arrow_forward1. For the following reaction, what is the rate law ? (image) 2. is the following nucleophile strong or weak HO^- a) strong b) weak c) not a nucleophile 3) what set of reaction conditions should favor an SN2 reaction on 2-bromo-3-methylbutane a) weak nucleophile in a protic solvent b) weal nucleophile in aprotic solvent c) strong nucleophile in a protic solvent d) strong nucleophile in a aprotic solventarrow_forwardDraw the structure of the starting material A for the reaction below when one equivalent of H2/Pt is present.arrow_forward
- What is an E2 mechanism? What are all of the steps, transition state, and all final products? What are the characteristics of a good base for E2? How does a chemist tell a strong versus weak or bulky versus not bulky nucleophile? Thank youarrow_forwardPlease complete boxed reactions. Thanks. P.s. this is not graded work but a study guide.arrow_forwardWhat are all three products that can result from this E1 mechanism? (1 major, 2 minor)?arrow_forward
- 1. What is the product for the first one and the mechanism for the second one?arrow_forwardDraw the products of the three step reaction sequence shown below. Ignore inorganic byproducts. If the reaction results in a mixture of ortho and para isomers, draw only the para-product. Select to Draw NO₂ 1. LiAlH4 2. H₂O* Cl₂ AICI 3 Select to Draw CH3C(O)CI Select to Drawarrow_forwardFor each reaction below identify the electrophile and the nucleophile.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

