
Concept explainers
(a)
Interpretation:
From the given compounds, compounds having delocalised electrons have to be identified.
Concept Introduction:
Delocalized electrons: Electrons that are not localized in one particular atom or bond and shared by three or more atoms are called delocalized electrons.
Resonance contributor: In some chemical compounds like benzene pi electrons are delocalized in it and difficult to locate it. Resonance contributor gives an idea of where about of pi electrons. The exact structure with localized electrons are called resonance contributor.
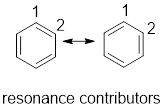
(a)
Interpretation:
From the given compounds, resonance contributor for compounds having delocalised electrons have to be drawn.
Concept Introduction:
Delocalized electrons: Electrons that are not localized in one particular atom or bond and shared by three or more atoms are called delocalized electrons.
Resonance contributor: In some chemical compounds like benzene pi electrons are delocalized in it and difficult to locate it. Resonance contributor gives an idea of whereabouts of pi electrons. The exact structure with localized electrons are called resonance contributor.
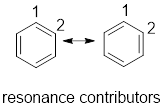
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Essential Organic Chemistry (3rd Edition)
- Add a + or above each curved arrow in Figure 4.11 to show the sign of the energy change.arrow_forwardFor each proposed set of resonance structures: a. (E) Add curved arrows (starting from left) to show how each successive r.s. was generated. b. Cross out any resonance structures that are NOT important, and explain your reasoning.arrow_forward(a) Add curved arrows to show how the starting material A is converted to the product B. (b) Draw all reasonable resonance structures for B. (c) Draw the resonance hybrid for B.arrow_forward
- Draw resonance contributors for the following species. Do not include structures that are so unstable that their contributions to the resonance hybrid would be negligible. Indicate which are major contributors and which are minor contributors to the resonance hybrid. b. Do any of the species have resonance contributors that all contribute equally to the resonance hybrid?arrow_forwardFor each of the following groups of molecules (labeled A–E), rank the resonance forms in order from least important (5) to most important (1)arrow_forwardCould you explain why the highlighted group on the right side of the double bond has a higher priority than the lower group on the right side of the double bond. I use this information to determine E/Z assigmentsarrow_forward
- Which of the following bonds absorbs higher energy? O--H or C--H? Why?arrow_forwardPleae match the choices from 'A' to 'S'arrow_forwardRizatriptan (trade name Maxalt) is a prescription drug used for the treatment of migraines. (a) How many aromatic rings does rizatriptan contain? (b) Determine the hybridization of each N atom. (c) In what type of orbital does the lone pair on each N reside? (d) Draw all the resonance structures for rizatriptan that contain only neutral atoms. (e) Draw all reasonableresonance structures for the ve-membered ring that contains three N atoms.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
