
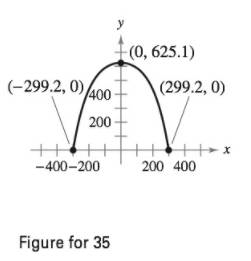
Length of Gateway Arch The Gateway Arch in St. Louis, Missouri, is closely approximated by the inverted catenary
Use the
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
Calculus: Early Transcendental Functions
- Van der Waals Equation In Exercise 18 at the end of Section 2.3, we discussed the ideal gas law, which shows the relationship among volume V, pressure p, and temperature T for a fixed amount 1 mole of a gas. But chemists believe that in many situations, the van der Waals equation gives more accurate results. If we measure temperature T in kelvins, volume V in liters, and pressure p in atmosphere 1 atm is the pressure exerted by the atmosphere at sea level, then the relationship for carbon dioxide is given by p=0.082TV0.0433.592V2atm What volume does this equation predict for 1 mole of carbon dioxide at 500 kelvins and 100 atm?Suggestion: Consider volumes ranging from 0.1 to 1 liter.arrow_forwarddefinite integrationarrow_forwarda) analytical method b) NEWTON-COTES CLOSED INTEGRATION FORMULA (PRECISE RULE)arrow_forward
- Algebra & Trigonometry with Analytic GeometryAlgebraISBN:9781133382119Author:SwokowskiPublisher:Cengage
 Functions and Change: A Modeling Approach to Coll...AlgebraISBN:9781337111348Author:Bruce Crauder, Benny Evans, Alan NoellPublisher:Cengage Learning
Functions and Change: A Modeling Approach to Coll...AlgebraISBN:9781337111348Author:Bruce Crauder, Benny Evans, Alan NoellPublisher:Cengage Learning

