
Concept explainers
a) -CH3, -OH, -H, -Cl
Interpretation:
The substituents in the set, -CH3, -OH, -H, -Cl, are to be ranked according to the sequence rules.
Concept introduction:
The member that ranks higher can be determined by considering the
To rank:
The substituents in the set, -CH3, -OH, -H, -Cl, according to the sequence rules.
b) -CH3, -CH2CH3, -CH=CH2, -CH2OH
Interpretation:
The substituents in the set, -CH3, -CH2CH3, -CH=CH2, -CH2OH, are to be ranked according to the sequence rules.
Concept introduction:
The member that ranks higher can be determined by considering the atomic number of the first atom in each substituent. The atom with highest atomic number gets the higher rank. If a decision cannot be made by considering the atomic number of the first atom in each substituent then the second, third, fourth atoms can be considered until the first difference is found. Multiple bonded atoms are considered as equivalent to the same number of single bonded atoms.
To rank:
The substituents in the set, -CH3, -CH2CH3, -CH=CH2, -CH2OH, according to the sequence rules.
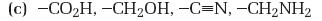
Interpretation:
The substituents in the given set are to be ranked according to the sequence rules.
Concept introduction:
The member that ranks higher can be determined by considering the atomic number of the first atom in each substituent. The atom with highest atomic number gets the higher rank. If a decision cannot be made by considering the atomic number of the first atom in each substituent then the second, third, fourth atoms can be considered until the first difference is found. Multiple bonded atoms are considered as equivalent to the same number of single bonded atoms.
To rank:
The substituents in the given set according to the sequence rules.
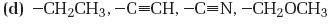
Interpretation:
The substituents in the given set are to be ranked according to the sequence rules.
Concept introduction:
The member that ranks higher can be determined by considering the atomic number of the first atom in each substituent. The atom with highest atomic number gets the higher rank. If a decision cannot be made by considering the atomic number of the first atom in each substituent then the second, third, fourth atoms can be considered until the first difference is found. Multiple bonded atoms are considered as equivalent to the same number of single bonded atoms.
To rank:
The substituents in the given set according to the sequence rules.
Trending nowThis is a popular solution!

Chapter 7 Solutions
Organic Chemistry
- Draw both cis- and trans-1,4-dimethylcyclohexane in their more stable chairconformations. (a) How many stereoisomers are there of cis-1,4-dimethylcyclohexane, and how many of trans-1,4-dimethylcyclohexane? (b) Are any of the structures chiral? (c) What are the stereochemical relationships among the various stereoisomers of 1,4-dimethylcyclohexane?arrow_forwardRank the following groups in order of decreasing priority. −H, −CH3, −Cl, −CH2Clarrow_forwardGiven the following partial structure, add a substituent X to C-1 so that it satisfies the indicated stereochemical requirement. What is the A -C - C - X torsion (dihedral) angle in each?arrow_forward
- Draw both cis- and trans-l,4-dimethylcyclohexane in their more stable chair conformations. a. How many stereoisomers are there of cis-1,4-dimethylcyclohexane, and how many of trans-l,4-dimethylcyclohexane? b. Are any of the structures chiral c. What are the stereochemical relationships among the various stereoisomers of 1,4-dimethylcyclohexane?arrow_forwardA 1,2-cis disubstituted cyclohexane, such as cis l,2-dichlorocyclohexane, musthave one group axial and one group equatorial. Explain.arrow_forwarddraw 6 acyclic constitutional isomers of C6H12. Provide a name for each isomer, inclduing E-,Z-,R,S where appropriatearrow_forward
- If I chlorinate 2-bromo-1-chloropropane under light, how many products do I obtain? Are any chiral?arrow_forwardShow how to convert cyclopentene into cis-1,2-Cyclopentanediolcompoundarrow_forward1.Draw both cis- and trans-1,4-dimethylcyclohexane in their conformations. (a) How many stereoisomérs are there of cis-1,4-dimethylcyclohexane, and how many of rans-1,4-dimethylcyclohexane? (b) Are of the structures chiral? (c) What are the stereochemicar relationships among the various stereoisomers of 1,4-dimethylcyclohexane?arrow_forward
- Order the following substituents from highest priority to lowest priority: CH3, OH, H,F. Use the Cahn-Ingold-Prelog priority system. a. F, CH3, OH, H b. H, CH3, F, OH c. F, OH, CH3, H d. H, CH3, OH, Farrow_forwardRings + Unsaturation --- Hydrogenation If compound A C51H81BrN5O3P3 is hydrogenated to give compound B C51H101BrN5O3P3. How many rings does compound A have? Assume that P has a valency of 5. Would the answer be 4 rings? Formula -> unsat + rings = 1+C +N/2 - H/2 - X/2arrow_forwardThe compounds drawn should each contain a cyclohexane ring. For all three compounds draw a wedge and dash structure, Chair I, and Chair II conformations. Formula: C9H18 with substitution 1,1- disubstituted with stereochemistry of (R,S) Formula: C7H13Cl with substitution 1,3- disubstituted with stereochemistry of (R,R) Formula: C7H14O with substitution 1,4- disubstituted with stereochemistry (S,S)arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
