
(a)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.
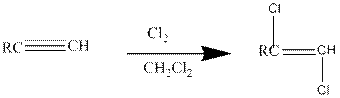
If excess halides are present, a second addition reaction also occurs.
(b)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
(c)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
(d)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
(e)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
(f)
Interpretation:
The major product of the given reaction should be determined.
Concept Introduction:
Addition of halides to alkynes:
Halogens like chlorine and bromine adds to the alkyne compounds. The general form of the reaction is given below.

If excess halides are present, a second addition reaction also occurs.
Want to see the full answer?
Check out a sample textbook solution
Chapter 7 Solutions
EBK ORGANIC CHEMISTRY
- For each reaction in question 20, sketch a reaction coordinate energy diagram indicating the starting materials, transition states and any intermediates, if present, in the reaction. Don't worry about the absolute energy of starting materials and products but you can assume that the product is more stable than the starting materials.arrow_forwardDetermine the major product of the following reaction OH H3O* H2 Pdarrow_forwardPlease provide the steps that each carbonation will undergo to form a carbonation that is more stable. Use arrows to explain the steps and provide the product for each.arrow_forward
- Draw structure of substitution product(s) & major elimination productarrow_forwardGive the major product of the reaction. CO,H 1) SOCI, 2) Cl2 3)A20arrow_forwardWhy won't most primary alkyl halides react in Friedel-Crafts alkylation reactions? O Primary alkyl halides undergo methyl shifts under the standard conditions of Friedel-Crafts alkylation reactions, making them unreactive. Primary alkyl halides create primary carbocations which are too high in energy and too readily undergo rearrangement. Primary alkyl halides do not form a sigma complex after a nucleophilic attack since they connect at the primary carbocation site. Primary alkyl halides as a reactant violates one of the three standard limitations to Friedel-Crafts alkylation reactions.arrow_forward
- Draw the major organic product for the reaction conditions shown. CH3 mCPBA CF3SO3H CH₂Cl₂arrow_forwardFor each of the following reactions predict the major product(s). LOCH3 CI AICI3 HNO3 NH2 1. NaNO2, HCI 2. HBF4, Aarrow_forwardGive the major product of the following reaction. ? 1) NaOH 2) CH;CH,CH,CI, There is no reaction under these conditions or the correct product is not listed here.arrow_forward
- pls also name the major product, thanks!arrow_forwardWhich of the following is the major product of the reaction shown? 1. LİAIH, 2. Н,О, Н* || CH,CH,CH,COH || HOCH,CH,COH HOCH,CH,CH,CH,0H CH,CH,CH,CH,OHarrow_forwardThe reaction of methylpropene with HBr in ether gives one of the two products below as the major product. Br HBr Br ether Product A Product B Product would have a higher energy transition state for the formation of the intermediate leading to it. O A O B O Both products would have the same transition state.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
