
Concept explainers
(a)
Interpretation: Check whether the given molecule is polar or not. If the molecule is polar then the direction of polarity has to be indicated.
Concept Introduction:
Polar molecules are the molecules having a positive and negative end and so there will be a charge separation.
A polar molecule is a molecule where the polar bonds are asymmetrically arranged (the dipoles do not cancel)
A nonpolar molecule is a molecule with no polar bonds or a molecule where the polar bonds are symmetrically arranged.
In polar molecule the charge separation occurred with respect to the difference in electronegativity of atoms in the molecule.
Direction of dipole moment in a molecule is can be represented as follows,

Different types of molecules and their geometry in accordance with the VSEPR theory are mentioned below,
(a)
Explanation of Solution
The structure of
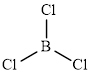
This molecule has a trigonal planar geometry. The direction dipole in the molecule is represented below,
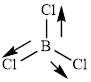
However
(b)
Interpretation: Check whether the given molecule is polar or not. If the molecule is polar then the direction of polarity has to be indicated.
Concept Introduction:
Polar molecules are the molecules having a positive and negative end and so there will be a charge separation.
A polar molecule is a molecule where the polar bonds are asymmetrically arranged (the dipoles do not cancel)
A nonpolar molecule is a molecule with no polar bonds or a molecule where the polar bonds are symmetrically arranged.
In polar molecule the charge separation occurred with respect to the difference in electronegativity of atoms in the molecule.
Direction of dipole moment in a molecule is can be represented as follows,

Different types of molecules and their geometry in accordance with the VSEPR theory are mentioned below,
(b)
Explanation of Solution
The structure of
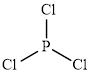
This molecule has a trigonal planar geometry. The direction dipole in the molecule is represented below,
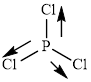
However
(c)
Interpretation: Check whether the given molecule is polar or not. If the molecule is polar then the direction of polarity has to be indicated.
Concept Introduction:
Polar molecules are the molecules having a positive and negative end (dipole) and so there will be a charge separation.
A polar molecule is a molecule where the polar bonds are asymmetrically arranged (the dipoles do not cancel)
A nonpolar molecule is a molecule with no polar bonds or a molecule where the polar bonds are symmetrically arranged.
In polar molecule the charge separation occurred with respect to the difference in electronegativity of atoms in the molecule.
Direction of dipole in a molecule is can be represented as follows,

Different types of molecules and their geometry in accordance with the VSEPR theory are mentioned below,
(c)
Explanation of Solution
The structure of
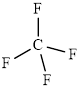
This molecule has a tetrahedral geometry. The direction dipole in the molecule is represented below,
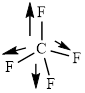
However
(d)
Interpretation: Check whether the given molecule is polar or not. If the molecule is polar then the direction of polarity has to be indicated.
Concept Introduction:
Polar molecules are the molecules having a positive and negative end and so there will be a charge separation.
A polar molecule is a molecule where the polar bonds are asymmetrically arranged (the dipoles do not cancel)
A nonpolar molecule is a molecule with no polar bonds or a molecule where the polar bonds are symmetrically arranged.
In polar molecule the charge separation occurred with respect to the difference in electronegativity of atoms in the molecule.
Direction of dipole moment in a molecule is can be represented as follows,

Different types of molecules and their geometry in accordance with the VSEPR theory are mentioned below,
(d)
Explanation of Solution
The structure of
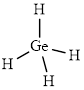
This molecule has a tetrahedral geometry. The direction dipole in the molecule is represented below,
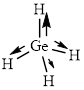
However
Want to see more full solutions like this?
Chapter 8 Solutions
Chemistry & Chemical Reactivity
- In each of the following molecules, a central atom is surrounded by a total of three atoms or unshared electron pairs: SnCl2, BCl3, SO2. In which of these molecules would you expect the bond angle to be less than 120? Explain your reasoning.arrow_forwardDifferentiate between covalent bonds and generallyweaker interactions such as polar covalent, hydrogen, andionic bonds.arrow_forwardThe C=O double bond is polar, with a dipole moment of 1.0 Debye. Do you expect CO2 to be a polar molecule? Why or why not?arrow_forward
- Consider the collection of nonmetallic elements O, P, Te,I, and B. (a) Which two would form the most polar singlebond? (b) Which two would form the longest single bond?(c) Which two would be likely to form a compound of formulaXY2? (d) Which combinations of elements would likelyyield a compound of empirical formula X2Y3?arrow_forwardHow many nonbonding electron pairs are there in each ofthe following molecules: (a) (CH3)2S, (b) HCN, (c) C2H2,(d) CH3F?arrow_forwardIf all of the electron groups around a single central atom are bonding, and the same outer atom is bonded to the central atom in each case, is the molecule polar, nonpolar, or does it depend on the identity of the outer atom? Explain.arrow_forward
- 1.4 PH3 and NH3 moleculea are the same shape but the molecules are non-polar and polar respectively. Explain why this is so.arrow_forwardHow many lone pairs of electrons are on the central atom of CHCI3 and what is the molecular shape?arrow_forward(a) Is the molecule BF3 polar or nonpolar? (b) If you react BF3to make the ion BF32-, is this ion planar? (c) Does the moleculeBF2Cl have a dipole moment?arrow_forward
 World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning



