
Concept explainers
Label the
a.![]() b.
b.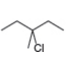 c.
c.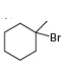
(a)
Interpretation:
Concept introduction: Carbon atom to which the halogen atom is attached is known as
Answer to Problem 8.1P
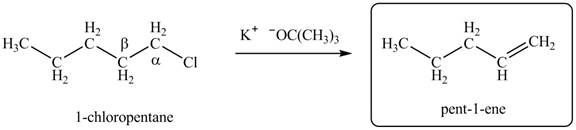
Explanation of Solution
There are different types of carbon atoms present in an alkyl halide depending on their relative position to the halogen atom.
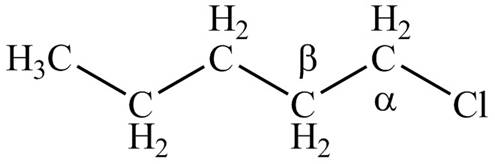
Figure 1
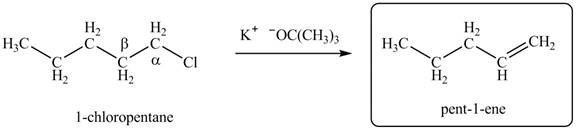
Figure 2
(b)
Interpretation:
Concept introduction: Carbon atom to which the halogen atom is attached is known as
Answer to Problem 8.1P
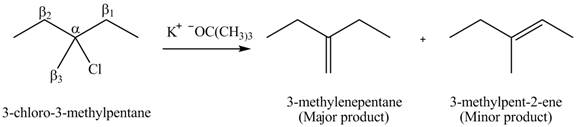
Explanation of Solution
There are different types of carbon atoms present in an alkyl halide depending on their relative position to the halogen atom.
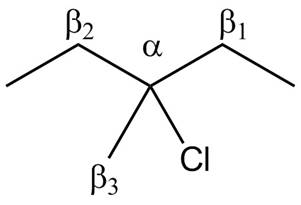
Figure 3
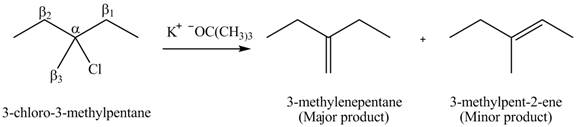
Figure 4
(c)
Interpretation:
Concept introduction: Carbon atom to which the halogen atom is attached is known as
Answer to Problem 8.1P
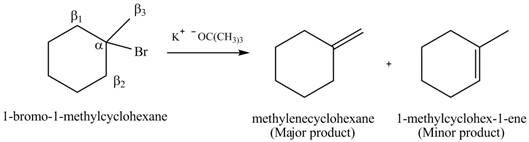
Explanation of Solution
There are different types of carbon atoms present in an alkyl halide depending on their relative position to the halogen atom.
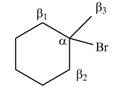
Figure 5
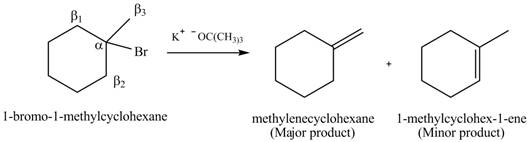
Figure 6
Want to see more full solutions like this?
Chapter 8 Solutions
KCTCS Organic Chemistry Value Edition (Looseleaf) - Text Only
- Label the α and β carbons in attached alkyl halide. Draw all possible elimination products formed when each alkyl halide is treated with K+−OC(CH3)3.arrow_forwardLabel the α and β carbons in each alkyl halide. Draw all possible elimination products formed when each alkyl halide is treated with K+−OC(CH3)3.arrow_forward7. GIVE AN EXAMPLE OF AN ENOL IN BIOLOGICAL SYSTEM 8. WHAT IS FEHLING'S TEST? WHAT ARE THE CONSTITUENTS OF FEHLING'S REAGENT? 10. HOW DOES FEHLING'S TEST COMPARE TO BENEDICT'S AND TOLLEN'S TEST?arrow_forward
- Ethers are oxidized with O2 to form hydroperoxides that decompose violently when heated. Draw a stepwise mechanism for this reaction.arrow_forwardLabel the a and b carbons in each alkyl halide. Draw all possible elimination products formed when each alkyl halide is treated with K+-OC(CH3)3.arrow_forwardWhat is the product formed in a-b when treated with one equivalent of HBr?arrow_forward
- Draw the products formed when A or B is treated with each reagent. In some cases, no reaction occurs.a. NaBH4, CH3OHb. [1] LiAlH4; [2] H2Oc. [1] CH3MgBr (excess); [2] H2Od. [1] C6H5Li (excess); [2] H2Oe. Na2Cr2O7, H2SO4, H2Oarrow_forward#20 B Draw structural formulas for all possible carbocations formed by the reaction of each alkene with HCl.arrow_forwardExplain Addition of Alcohols—Acetal Formation ?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





