
Concept explainers
Draw the structure of the conjugate base of each acid.
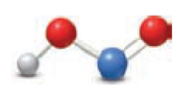
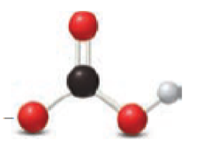
(a)
Interpretation:
The conjugate base of the acid
Concept Introduction:
Conjugate base: A conjugate base is the product formed by a loss of proton from an acid. The conjugate base of the acid A will be
Explanation of Solution
A proton is removed from the acid to form the conjugate base.
Nitrous acid loses a proton to form nitrite ion
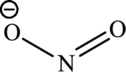
(b)
Interpretation:
The conjugate base of carbonic acid has to be drawn.
Concept Introduction:
Refer to part (a).
Explanation of Solution
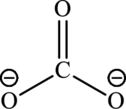
The conjugate base is formed by removing a proton from the acid.
Carbonic acid
Want to see more full solutions like this?
Chapter 8 Solutions
Principles of General, Organic, Biological Chemistry
- (E) Label each of the following as strong acid, strong base, or neither.arrow_forwardFor the previous four questions, label each molecule that appears in the question or your answer asstrong acid, strong base, weak acid, or weak base.arrow_forwardA very strong base can remove a proton from methylamine:arrow_forward
- Mark each of the following statements True or False: a. The conjugate base of a strong acid is always a weak base. b. The conjugate acid of a strong base is always a weak acid. c. The stronger the acid, the weaker its conjugate base, and vice versa.arrow_forwardAnswer true or false to the following statements about the mechanism of acid-base reactions. (a) The acid and base must encounter each other by a collision in order for the proton to transfer. (b) All collisions between acids and bases result in proton transfer. (c) During an acid-base reaction the lone pair on the base fills the A-H antibonding sigma orbital.arrow_forwardOne structure for the conjugate acid of acetone is The electrons in the carbon–oxygen double bond are pushable electrons, and the atom is a receptor.arrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning
World of Chemistry, 3rd editionChemistryISBN:9781133109655Author:Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCostePublisher:Brooks / Cole / Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





