
Concept explainers
(a)
Interpretation: The more reactive alkene towards the acid-catalyzed hydration is to be interpreted.
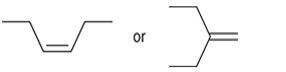
Concept introduction:
(b)
Interpretation: The more reactive alkene from 2-methyl-2-butene and 3-methyl-1-butene toward the acid-catalyzed hydration is to be interpreted.
Concept introduction:
Alkenes are unsaturated hydrocarbons with at least one double bond between the carbon atoms. The presence of pi bonds in these molecules makes them more reactive compared to saturated hydrocarbons; alkanes. The stability of alkenes depends on the substituted groups.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
ORGANIC CHEMISTRY-WILEYPLUS NEXTGEN
- Write the structural formula of the organic product for the given reaction between an alkyne and an alkyl halide. The alkyne group is shown and should be entered as "CC" without the triple bond. Enter CC before associated HH atoms (e.g., CH3CH2CH2OCHCHCH3). what is the name of the product. 1.NaNH2 CH3CH2CCH------> product 2. CH3CH2Brarrow_forward6. Give the reagent and the reaction conditions that would distinguish between the following compounds. Explain how the reagent or condition would react with each compound, you may use the chemical equation for the reactions involved. a. phenol and isopentyl alcohol b. tert-butyl alcohol and isobutyl alcohol c. neopentyl alcohol and ether d. sec-butyl alcohol and neopentyl alcohol e. propene and butanolarrow_forwardChemistry 4. Which of the following reactions would produce the following alkene in a reasonable percentage yield, i.e., greater than 50%? HS0. hect OH CH,Br Ngl celone a. I b. II c. III d. More than one of these reactions e. None of these reactionsarrow_forward
- Choose the major product when 2-bromo-2-methylpentane is treated with sodium ethoxide in ethanol a.(E)-4-methylpent-2-ene b.2-methylpent-2-ene c.(Z)-4-methylpent-2-ene d.2-methylpent-1-enearrow_forward2. Which of the following alkynes will form only 1 product in acid-catalyzed hydration? a. 2-pentyne b. 3-hexyne c. 3-pentyne d. 1-hexynearrow_forward6. Give the structures of E2 reaction products of the following alkyl halides with a strong base. a a. 11 CI b. III Clarrow_forward
- Write the structural formula of the organic product for the given reaction between an alkyne and an alkyl halide. The alkyne group is shown and should be entered as "CC" without the triple bond. Enter CC before associated HH atoms (e.g., CH3CH2CH2OCHCHCH3). 1.NaNH2 CH3CH2CCH------> 2. CH3CH2b2arrow_forwardThe acid base reaction below is an example of a Lewis acid/base. In the boxes below each reagent indicate whether it is a Lewis Acid or a Lewis Base. The product of this reaction will have formal charges on the atoms indicated. Give the formal charge for these atoms in the attached boxes `NH2 `NH2 Rank the following alkene based on stability: a. b. d.arrow_forwardOH CH, -CH3 2. CH3-CH2-C-CH,-CH3 H+ CH,-CH, 3. Write structural formulas for all the alkene products that are formed under the indicated reaction conditions: (each is a synthesis problem, not a mechanism to show like the previous questions.) a. 1-methylcyclohexyl chloride (sodium ethoxide-a strong base, ethanol) b. 3-iodo-2,4-dimethylpentane (sodium ethoxide, ethanol)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





