
(a)
Interpretation: In the oxymercuration-demercuration reaction, a nucleophile other than water can be used. In the given reaction mechanism, the product is to be interpreted.
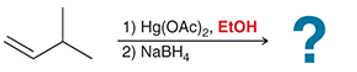
Concept introduction:
Addition reactions are the
(b)
Interpretation: In the oxymercuration-demercuration reaction, a nucleophile other than water can be used. In the given reaction mechanism, the product is to be interpreted.
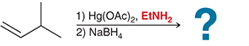
Concept introduction:
Addition reactions are the chemical reactions that show the addition of a certain group or molecule to the unsaturated carbon atoms. Alkenes are the unsaturated hydrocarbon that contains at least one double bond between the carbon atoms. Therefore, they tend to give addition reactions. Some examples of addition reactions are acid-catalyzed hydration, oxymercuration-demercuration, etc.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
ORGANIC CHEMISTRY
- a) Consider the reaction of HBr with ethylene and propylene. At roomtemperature the reaction of propylene with HBr is much faster than thereaction with ethylene.Using reaction energy diagrams and your knowledge of carbocationstability explain why this is so. b) Xylene (dimethylbenzene) is a commonly used chemical in the printingindustry and as a cleaning solvent for oily waste. It is also used whenpreparing histological samples to remove waxes from biological samples.Draw the three possible structures for this compound and give the UPACnames for each. Define which structures are ortho, meta, and para.arrow_forwardIn an electrophilic aromatic substitution, the substitution pattern on the product is determine by the directing nature of the substituents on the ring. What controls the substitution pattern in a nucleophilic aromatic substitution reaction?arrow_forwardFill both dashes in the paragraph Compounds containing a phenol group may work as ANTIOXIDANTS to prevent free radical damage. This is accomplished when a free radical (or UV light) encounters a phenol group, turning the phenol group into a radical. However, contrary to typical radical behavior, the structure of the phenol radical can neutralize (or quench) the unpaired electron. Specifically, the phenol structure neutralizes (or quenches) the unpaired radical electron by doing the following: taking the electron and ---------. The correct name (or abbreviation) of an example compound (discussed in the lecture videos) containing a phenol group with antioxidant properties is: ---------.arrow_forward
- An electron-deficient carbon atom reacts with a nucleophile, symbolized as: Nu−. Define this ?arrow_forwardWhich reaction or statement regarding nucleophilic substitutions is incorrect? A) C₁ + 2 H2O ноттон + 2 HCI B) ta CI+MeOH to + HCI C) D) The rate-limiting step in SN1 reactions is the initial step, loss of the leaving group. Nucleophilic substitution reactions that follow second-order kinetics involve complete inversion of configuration.arrow_forwardHeterolysis of the C–Z bond can generate a carbocation or a carbanion. Explain How ?arrow_forward
- Only one of these statements about nucleophilic aromatic substitution is true. O as the number of ortho and para electron withdrawing groups increases, the reaction rate decreases O the reaction proceeds by an addition-elimination mechanism the reaction involves a carbocation intermediate with delocalization of electrons all are falsearrow_forwardAll of the following are true of SN2 reactions except: The rate of the SN2 reaction of a substrate and a nucleophile is the same as the rate of the E2 reaction of the same two compounds. The rate varies with the concentration of nucleophile The rate varies with the type of nucleophile The nucleophile is involved in the rate-determining step Type: MCarrow_forward46. The Strength of Nucleophile: Which one is the strongest nucleophile? OH (B) 0-CH, -CH, (D) O (C) O (A) O (D) O (B)arrow_forward
- Compounds containing a phenol group may work as ANTIOXIDANTS to prevent free radical damage. This is accomplished when a free radical (or UV light) encounters a phenol group, turning the phenol group into a radical. However, contrary to typical radical behavior, the structure of the phenol radical can neutralize (or quench) the unpaired electron. Specifically, the phenol structure neutralizes (or quenches) the unpaired radical electron by doing the following: taking the electron and The correct name (or abbreviation) of an example compound (discussed in the lecture videos) containing a phenol group with antioxidant properties is:arrow_forward. One of these reactions occurs rapidly while the other is so slow that substitution products are not observed. Determine which reaction is which and explain the difference in rate using structural drawing and a few words. Br + 'Br +arrow_forwardFill the blank space. Compounds containing a phenol group may work as ANTIOXIDANTS to prevent free radical damage. This is accomplished when a free radical (or UV light) encounters a phenol group, turning the phenol group into a radical. However, contrary to typical radical behavior, the structure of the phenol radical can neutralize (or quench) the unpaired electron. Specifically, the phenol structure neutralizes (or quenches) the unpaired radical electron by doing the following: taking the electron and ---------. The correct name (or abbreviation) of an example compound containing a phenol group with antioxidant properties is: ---------.arrow_forward
 Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
Chemistry for Today: General, Organic, and Bioche...ChemistryISBN:9781305960060Author:Spencer L. Seager, Michael R. Slabaugh, Maren S. HansenPublisher:Cengage Learning
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


