
(a)
Interpretation: The hydroboration-oxidation reaction product of alkene is to be interpreted.
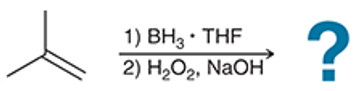
Concept introduction:
Addition reactions are the
(b)
Interpretation: The hydroboration-oxidation reaction product of alkene is to be interpreted.
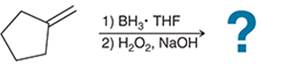
Concept introduction:
Addition reactions are the chemical reactions that show the addition of a certain group or molecule to the unsaturated carbon atoms. Alkenes are the unsaturated hydrocarbon that contains at least one double bond between the carbon atoms. Therefore, they tend to give addition reactions. Some examples of addition reactions are acid-catalyzed hydration, hydroboration-oxidation, oxymercuration-demercuration, etc.
(c)
Interpretation: The hydroboration-oxidation reaction product of alkene is to be interpreted.
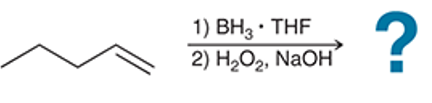
Concept introduction:
Addition reactions are the chemical reactions that show the addition of a certain group or molecule to the unsaturated carbon atoms. Alkenes are the unsaturated hydrocarbon that contains at least one double bond between the carbon atoms. Therefore, they tend to give addition reactions. Some examples of addition reactions are acid-catalyzed hydration, hydroboration-oxidation, oxymercuration-demercuration, etc.
Want to see the full answer?
Check out a sample textbook solution
Chapter 8 Solutions
ORGANIC CHEMISTRY
- 6A Give the major products in the following reactions and explain CN (i) KMnO4 (ii) T. H₂SO4arrow_forwardGive answer all questions with explanationarrow_forwardDescribe the species that would result from the subsequent transfer of the proton from the metal to one of the Cp rings of ferrocene. Give the formal oxidation state of the metal centre and the valence electron countarrow_forward
- Show Calculations and Formulas. What is the energetic favorability of A? (A) MnO2 reduction by sulfide yielding S0 Compared to the energetic favorability of B? (B) energetic favorability of MnO2 reduction by sulfide yielding SO42-.arrow_forwardChemistry Assuming that the reaction of E,E-2,4-hexadien-1-ol with maleic anhydride falls in the“normal electron demand” classification, illustrate the molecular orbitals involved in this reaction and how they will react.arrow_forwardWhat products are expected from the oxidation with hot KMnO4 (a) solution of (a) PGE1, and (b) PGE1α?arrow_forward
- (a) What is the product of the following reaction? What is the catalytically active Pd species, and how is it formed? Draw a catalytic cycle, explaining what type of reaction is occurring at each stage. Indicate the formal oxidation state of Pd for each intermediate in the catalytic cycle, state how many valence electrons each intermediate has, and indicate the geometry of each Pd complex. Why is LiCl necessary? OTf OMe + Me. SnBu3 cat. PdCl₂(PPh3)2 NEt3, LICI, THEarrow_forward10arrow_forwardConsider the following reaction (DPPE structure below): B(OH)2 F. Ni(OTf)2 DPPE K2CO3 (a) Give the product of the reaction and suggest a plausible catalytic cycle giving the number of d-electrons and the oxidation state of Ni in each intermediate.arrow_forward
- 4arrow_forwardPredict the major product of the following reactions..Include stereochemistry where appropriate – for all chiral products, indicate whether they are racemic or nonracemic.arrow_forwardConsider a hypothetical chemical reaction between compound A and compound B, which produces compound C as the final product. The reaction is known to be exothermic and spontaneous. However, when the reaction is carried out under certain conditions, it fails to occur. Explain this observation and propose a potential solution to overcome this hurdle.arrow_forward
