
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Question
Chapter 8.9, Problem 8.66P
Interpretation Introduction
Interpretation:
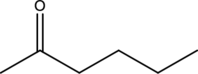
Preparation of the given compound from ethyl acetoacetate has to be given.
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Show how the following compounds can be synthesized. The only carbon-containing compounds available to you for each synthesis are shown.
How would you synthesize the methyl ketone shown below via the acetoacetic ester synthesis? You must show the reactions/reagents used in their correct order
Show how you would synthesize the following esters from appropriate acyl chloridesand alcohols. (c) benzyl benzoate (d) cyclopropyl cyclohexanecarboxylate
Chapter 8 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.1 - For each of the compounds below, identify all...Ch. 8.2 - Prob. 8.9PCh. 8.2 - Prob. 8.10PCh. 8.2 - Prob. 8.11PCh. 8.2 - Prob. 8.12P
Ch. 8.2 - Prob. 8.13PCh. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.3 - Predict the products of each of the following...Ch. 8.4 - Prob. 8.20PCh. 8.4 - Prob. 8.21PCh. 8.4 - Prob. 8.22PCh. 8.4 - Prob. 8.23PCh. 8.5 - Prob. 8.25PCh. 8.5 - Prob. 8.26PCh. 8.5 - On a separate piece of paper, draw a mechanism for...Ch. 8.6 - Prob. 8.29PCh. 8.6 - Predict the major product for each of the...Ch. 8.6 - Predict the major product for each of the...Ch. 8.6 - Predict the major product for each of the...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.6 - Identify the reagents you would use to achieve...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Predict the major product for each of the...Ch. 8.7 - Prob. 8.42PCh. 8.7 - Prob. 8.43PCh. 8.7 - Prob. 8.44PCh. 8.7 - Prob. 8.45PCh. 8.7 - Prob. 8.47PCh. 8.7 - Prob. 8.48PCh. 8.7 - Prob. 8.49PCh. 8.7 - Prob. 8.50PCh. 8.8 - Prob. 8.52PCh. 8.8 - Prob. 8.53PCh. 8.8 - Prob. 8.54PCh. 8.8 - Prob. 8.55PCh. 8.8 - Prob. 8.57PCh. 8.8 - Prob. 8.58PCh. 8.8 - Prob. 8.59PCh. 8.8 - Prob. 8.60PCh. 8.8 - Propose a mechanism for each of the following...Ch. 8.8 - Propose a mechanism for each of the following...Ch. 8.8 - Prob. 8.64PCh. 8.9 - Prob. 8.66PCh. 8.9 - Prob. 8.67PCh. 8.9 - Prob. 8.68PCh. 8.9 - Prob. 8.69PCh. 8.9 - Prob. 8.70PCh. 8.9 - Prob. 8.71PCh. 8.9 - Prob. 8.72PCh. 8.9 - Identify what reagents you would use to achieve...Ch. 8.9 - Identify what reagents you would use to achieve...Ch. 8.9 - Identify what reagents you would use to achieve...Ch. 8.10 - Prob. 8.78PCh. 8.10 - Prob. 8.79PCh. 8.10 - Prob. 8.80PCh. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...Ch. 8.10 - Propose a synthesis for each of the following...
Knowledge Booster
Similar questions
- How would you prepare the following ketones using an acetoacetic ester synthesis?arrow_forwardShow how you would synthesize the following esters from appropriate acyl chloridesand alcohols.(a) ethyl propionate (b) phenyl 3-methylhexanoate(c) benzyl benzoate (d) cyclopropyl cyclohexanecarboxylate(e) tert-butyl acetatearrow_forwardWhat is the mechanism and product when the following molecule is treated with H2SO4 and acetone?arrow_forward
- Show the product expected when the following unsaturated -ketoester is treated with each reagent. (a) Pd,EtOHH2(1mol) (b) CH3OHNaBH4 (c) 2.H2O1.LiAlH4,THE (d) 2.H2O1.DABALH,78arrow_forwardShow how you would synthesize the following esters from appropriate acyl chloridesand alcohols.(a) ethyl propionate (b) phenyl 3-methylhexanoatearrow_forwardWhat is the reaction mechanism for ethyl 2,4-diphenylacetoacetate by a Claisen condensation with ethyl phenylacetate?arrow_forward
- Show how you would convert aniline to the following compounds.( b) chlorobenzenearrow_forwardWhich substrate will you use to synthesize 1-propanol from propanal? A.) NaBH₄ B.) O₃ C.) KMnO₄ D.) CH₃MgCl E.) None of the given answerarrow_forwardShow how you would accomplish the following syntheses. ) hexan@1@ol S 2-hydroxyheptanoic acidarrow_forward
- Esters with only one α hydrogen generally give poor yields in the Claisen condensation. Q: Propose a mechanism for the Claisen condensation of ethyl isobutyrate.arrow_forwardShow the resonance forms for the enolate ions that result when the following compoundsare treated with a strong base.(a) ethyl acetoacetate (b) pentane-2,4-dione(c) ethyl a@cyanoacetate (d) nitroacetonearrow_forwardPredict the outcome of the intramolecular aldol condensation of the following compounds:arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning