
Interpretation:
A balanced equation for the given hypothetical reaction is to be written, and the oxidation and hybridization state of sulfur in
required and the mass of
produced is to be calculated.
Concept introduction:
The oxidation state of an element is zero. The sum of the oxidation states of all the elements is equal to zero in a molecule of a compound, and in case of an ion, it is equal to the charge on the ion. Oxygen has a fixed oxidation state of
in its compounds.
To find hybridisation of an atom in a molecule, at first draw the Lewis structure of the molecule. Find the number of electrons domains around the atom. This gives the number of hybrid orbitals required for bonding. The number of hybrid orbitals is equal to the number of atomic orbitals that hybridise. Thus, one
and one
orbital hybridize to form two
and two
orbitals hybridize to form three
hybrid orbitals, and one
and three
orbitals hybridize to form four
hybrid orbitals.
The conversion factor is a fraction that is used to convert one unit to another. Use of more than one factor to find a solution is called dimensional analysis.
Answer to Problem 115AP
Solution: The balanced equation for the reaction is as follows:
The oxidation state of sulfur in elemental sulfur
is
is
is
is
is
and mass of
produced is
Explanation of Solution
The hypothetical reaction between elemental sulfur and sulfur trioxide is as follows:
The balanced equation for this reaction is as follows:
In
The oxidation state of oxygen is
Let
is the oxidation state of sulfur.
Then
is calculated as follows:
The oxidation state of sulfur in
is
In
Let
is the oxidation state of sulfur and the oxidation state of oxygen is
is calculated as follows:
The oxidation state of sulfur in
is
The Lewis structure of
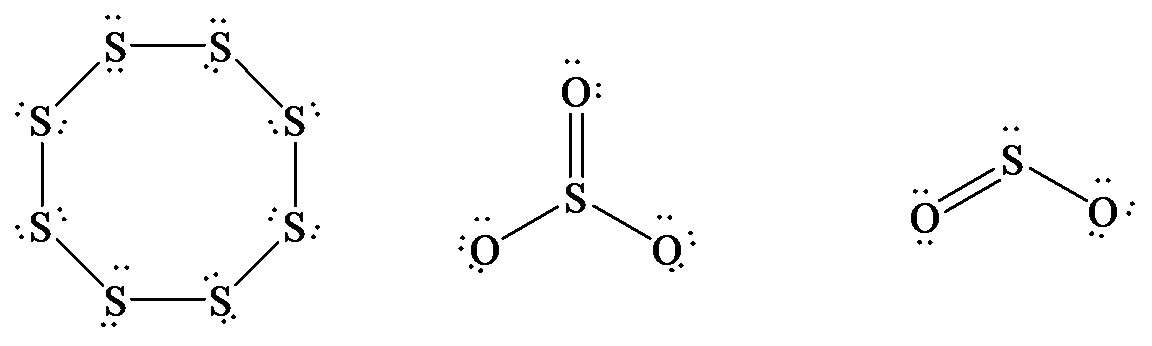
In
hybrid orbitals.
In
hybrid orbitals.
In
hybrid orbitals.
Consider the balanced equation:
One mole of
combines with
moles of
moles of
is
is given as
Convert the mass of
to moles as follows:
One mole of
combines with
moles of
Thus, for
needed is calculated as follows:
Molar mass of
is
Thus, the amount of
One mole of
produces
moles of
Thus, for
produced is calculated as follows:
Molar mass of
is
Thus, the amount of
Hence, mass of
required is
produced is
The balanced equation for the reaction between elemental sulfur and sulfur trioxide to produce sulfur dioxide is written; the oxidation and hybridization state of sulfur in
required and the mass of
produced by
of elemental sulfur are calculated.
Want to see more full solutions like this?
Chapter 9 Solutions
Chemistry
- In addition to NF3, two other fluoro derivatives of nitrogen are known: N2F4 and N2F2. What shapes do you predict for these two molecules? What is the hybridization for the nitrogen in each molecule?arrow_forwardIn propene CH3CH=CH2, the first carbon has sp3 hybrid orbitals and the second carbon has sp2 hybrid orbitals. These orbitals interact to make a bond. Why are these hybrid orbitals not orthogonal?arrow_forwardConsider the polyatomic ion IO65-. How many pairs of electrons are around the central iodine atom? What is its hybridization? Describe the geometry of the ion.arrow_forward
- The sulfamate ion, H2NSO3, can be thought of as having been formed from the amide ion, NH2, and sulphur trioxide, SO3. (a) What are the electron-pair and molecular geometries or the amide ion and or SO3? What are the hybridizations of the N and S atoms, respectively? (b) Sketch a structure for the sulfamate ion, and estimate the bond angles. (c) What changes in hybridization do you expect for N and S in the course of the reaction NH2 + SO3 H2NSO3? (d) Is SO3 the donor of an electron pair or the acceptor of an electron pair in the reaction with amide ion? Does the electrostatic potential map shown below confirm your prediction?arrow_forwardDo lone pairs about a central atom affect the hybridization of the central atom? If so, how?arrow_forwardWhat modification to the molecular orbital model was made from the experimental evidence that B2 is paramagnetic?arrow_forward
- The structure of amphetamine, a stimulant, is shown below. (Replacing one H atom on the NH2, or amino, group with CH3 gives methamphetamine a particularly dangerous drug commonly known as speed.) (a) What are the hybrid orbitals used by the C atoms of the C6 ring. by the C atoms of the side chain, and by the N atom? (b) Give approximate values for the bond angles A, B, and C. (c) How many bonds and bonds are in the molerule? (d) Is the molecule polar or nonpolar? (e) Amphetamine reacts readily with a proton (H+) in aqueous solution. Where does this proton attach to the molecule? Explain how the electrostatic potential map predicts this site of protonation.arrow_forwardCinnamaldehyde ocaus naturally in cinnamon oil. (a) What is the most polar bond in the molecule? (b) How many bonds and how many bonds are there? (c) Is cis-trans isomerism possible? If so, draw the isomers of the molecule. (d) Give the hybridization of the C atoms in the molecule. (e) What are the values of the bond angles 1, 2, and 3 ?arrow_forwardThe compound sketched below is acetylsalicylic acid, commonly known as aspirin. (a) What are the approximate values of the angles marked A, B, C, and D? (b) What hybrid orbitals are used by carbon atoms 1, 2, and 3ss?arrow_forward
- Considering only the molecular orbitals formed by combinations of the 2s atomic orbitals, how many molecular orbitals can be formed by 1000 Li atoms? In the lowest energy state, how many of these orbitals will be populated by pairs of electrons and how many will be empty?arrow_forwardWhat hybrid orbitals would be expected for the central atom in each of the following molecules or ions?arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





