Fluid Pressure
The term fluid pressure is coined as, the measurement of the force per unit area of a given surface of a closed container. It is a branch of physics that helps to study the properties of fluid under various conditions of force.
Gauge Pressure
Pressure is the physical force acting per unit area on a body; the applied force is perpendicular to the surface of the object per unit area. The air around us at sea level exerts a pressure (atmospheric pressure) of about 14.7 psi but this doesn’t seem to bother anyone as the bodily fluids are constantly pushing outwards with the same force but if one swims down into the ocean a few feet below the surface one can notice the difference, there is increased pressure on the eardrum, this is due to an increase in hydrostatic pressure.
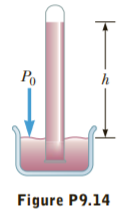
Blaise Pascal duplicated Torricelli’s barometer using a red Bordeaux wine, of density 984 kg/m3 as the working liquid (Fig. P9.14). (a) What was the height h of the wine column for normal atmospheric pressure? (b) Would you expect the vacuum above the column to be as good as for mercury?
Trending now
This is a popular solution!
Step by step
Solved in 3 steps with 2 images









