
Concept explainers
(a)
Interpretation:
cis isomer should be drawn for the given trans isomer.
Concept Introduction:
The arrangement of groups attached to the carbo atoms involved in a
Cis Isomer: The two groups attached on same side of
In cis isomer the hydrogen atoms are on the same side of the two carbon atom.
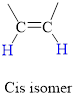
Trans isomer: The two groups are attached on opposite sides of the carbon-carbon double bond;.
The hydrogen atoms are on the opposite side of the two carbon atom.
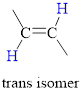
(b)
Interpretation:
trans isomer should be drawn for the given cis isomer.
Concept Introduction:
The arrangement of groups attached to the carbo atoms involved in a
Cis Isomer: The two groups attached on same side of
In cis isomer the hydrogen atoms are on the same side of the two carbon atom.

Trans isomer: The two groups are attached on opposite sides of the carbon-carbon double bond;.
The hydrogen atoms are on the opposite side of the two carbon atom.

Want to see the full answer?
Check out a sample textbook solution
Chapter 9 Solutions
Chemistry & Chemical Reactivity
- Which of the following central atoms would be expected to accommodate more than four electron pairs in some of its compounds?Simply explain your answer in a seperate sheet as well. F Br O Narrow_forwardWhat exactly is the difference between (E)/(Z) and cis/trans nomenclature? In my class, we usually use (E)/(Z) for double bonds- is it possible to use it for single bonds? Is (E)/(Z) used when cis/trans is not able to depict the geometric bonding as there are four different groups?arrow_forwardAccording to Werner's postulate, the sum of the primary and secondary valences in the compound hexaaminecobalt(III) oxalate is... A. 3 and 2 B. 3 and 6 C. 2 and 3 D. 2 and 6arrow_forward
- Draw all the isomers of C4H10O using bond-line formulas.arrow_forwardWhich compound of the following pairs would you expect to have the greatest dipole moment and by what? Draw its electrostatic potential map (without colors), indicating the direction of polarization of each link, using the convention δ + / δ- a) LiH or KH b) CH3NH2 or SiBr4arrow_forwardHow many isomers of all kinds the [Co(NH3)2(H2O)2(OH)2]+ has?arrow_forward
- Consider the compound C₂H₃N. Which one of the structures in Figure 4 is the best representation of this compound based on your current knowledge? * A B C D All these structures are good representations of the compound.arrow_forwardWhich of the compounds below are cis/trans isomerism applicable? Choices: A. 2 onlyB.2 and 3C. all 3D. 1 and 2E. 3 onlyarrow_forwardWrite Lewis structures for the Cis -trans isomers of CH3CH = CHCl.arrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning





