
Concept explainers
RIPs as Cancer Drugs Researchers are taking a page from the structure-function relationship of RIPs in their quest for cancer treatments. The most toxic RIPs, remember, have one domain that interferes with ribosomes, and another that carries them into cells. Melissa Cheung and her colleagues incorporated a peptide that binds to skin cancer cells into the enzymatic part of an RIP, the E. coli Shiga-like toxin. The researchers created a new RIP that specifically kills skin cancer cells, which are notoriously resistant to established therapies. Some of their results are shown in FIGURE 9.17.
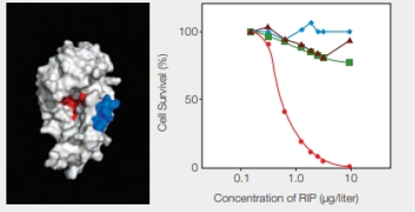
FIGURE 9.17 Effect of an engineered RIP on cancer cells. The model on the left shows the enzyme portion of E. coli Shiga-like toxin engineered to carry a small sequence of amino acids (in blue) that targets skin cancer cells. (Red indicates the active site.) The graph on the right shows the effect of this engineered RIP on human cancer cells of the skin ( ); breast (
); breast ( ) liver (
) liver ( ); and prostate (
); and prostate ( ).
).
Which cells survived best at 1 microgram per liter RIP?
Trending nowThis is a popular solution!

Chapter 9 Solutions
Biology: The Unity and Diversity of Life (MindTap Course List)
Additional Science Textbook Solutions
Human Physiology: An Integrated Approach (7th Edition)
Biology: Concepts and Investigations
Microbiology Fundamentals: A Clinical Approach - Standalone book
Human Anatomy & Physiology (2nd Edition)
BIOLOGY:THE ESSENTIALS (LL) W/CONNECT
- Polypeptide folding is often mediated by other proteins called chaperones. Describe how a mutant chaperone protein might be responsible for a genetic disorder involving an enzyme.arrow_forwardUnderstanding the Relevance of Chaperones in Protein Folding Protein molecules, like all molecules, can be characterized in terms of general properties such as size, shape, charge, solubility/hydrophobicity. Consider the influence of each of these general features on the likelihood of whether folding of a particular protein will require chaperone assistance or not. Be specific regarding just Hsp7O chaperones or Hsp7O chaperones and Hsp60 chaperonins.arrow_forwardExploring the Structure of the 30S Ribosomal Subunit Go to www.pdh.org and bring up PDB file 1GIX, which shows the 30S ribosomal subunit, the three tRNAs, and mRNA. In the box on the right titled ‘Biological Assembly.� click “More Images.� and then scroll down to look at the Interactive Vic By moving your cursor over the image, you can rotate it to view it from any perspective. a. How are the ribosomal proteins represented in the image? b. How is the 16S rRNA portrayed? c. Rotate the image to see how the tRNAs stick out from the structure. Which end of the tRNA is sticking out? d. Where will these ends of the tRNAs lie when the 50S subunit binds to this complex?arrow_forward
- What forces come into play with protein folding (please explain this at the molecular level)?arrow_forwardLoop regions play important roles in the secondary structure of protein. Define loop region and give three (3) of the rolesarrow_forward3) You are working on a protein with the following sequence in an area of interest. -Asp-Leu-Leu-Gln-Glu-Glu-Asp-Glu-Ser-Arg a. The current structure, solved at pH 7.4, of this segment has an alpha helix that is disrupted a er the Gln. Why might the alpha helix stop at this residue? b. This protein is involved in Lysosomes in vivo. The secondary structure of this region is expected to change into a complete alpha helix. Why might this change into a complete helix? ( Hint: Lysosomes are acidic!)arrow_forward
- Full length resilin that is not in a cell is thought by some to form a tertiary structure of this form (picture attached) The yellow portions represent exon 1 and the blue portions exon 3. These structures assemble into a kind of lattice or network. In full length resilin, stress in the form of mechanical pressure or heating to remove water results in a tighter network that excludes any water internally. How might this behavior of resilin produce its ‘elasticity’? Explain how the looser water containing structure and the tighter water excluding structure may give us a mechanism for the elasticity.arrow_forwardIn the early days of research on protein synthesis, some scientists observed that their most highly purified ribosome preparations, containing almost exclusively single ribosomes, were less active than preparations that were less highly purified. Suggest an explanation for this observation.arrow_forwardProtein Structure and Function A common strategy in the regulation of protein function is to alter its structure. Describe two specific strategies used by the cell to alter a protein’s structure, thereby altering its function.arrow_forward
- To better understand the effects of palmitoylation on protein X, researchers want to chemically attach a palmitic acid at a specific position in this protein. The researchers first try to feed cells with palmitic acid or an amino acid with a C16 side chain. Neither of the initial experiments is successful in helping the researchers learn more about the biological role of palmitoylated protein X. Why? Suggest a strategy to specifically incorporate a palmitoylation site into protein X.arrow_forwardAnfinsen studied protein folding using RNaseA. In one experiment we didn’t discuss in class, he heated RNase in the presence of BME, then he transferred the protein out of water into benzene, and cooled the protein in the presence of O2. Under these conditions, did Anfinsen find the protein was active? Justify your answer.arrow_forwardUsing protein leptins' primary, secondary, and tertiary structure, explain your understanding on their differences. If you happen to mutate (change) the amino acid(s), then write or draw its possible primary, secondary and tertiary structure. Explain and compare this to the original structure of this protein.arrow_forward
 Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning
Biology: The Unity and Diversity of Life (MindTap...BiologyISBN:9781305073951Author:Cecie Starr, Ralph Taggart, Christine Evers, Lisa StarrPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning
Human Heredity: Principles and Issues (MindTap Co...BiologyISBN:9781305251052Author:Michael CummingsPublisher:Cengage Learning


