
(a)
Interpretation:
The most stable carbocation structural formula of given molecular formula has to be drawn.
Concept Introduction:
The most stable carbocation structural formula:
The most stable structural arrangement of atoms in a carbocation molecule is known as most stable carbocation structural formula.
The highly alkyl substituted carbocation is more stable
Hence, the stability of carbocation is,
(a)
Answer to Problem 9.10P
The most stable carbocation structural formula of given molecular formula is,
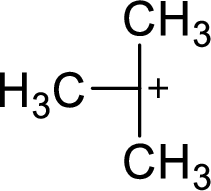
Explanation of Solution
The highly alkyl substituted carbocation is more stable. Hence, the most stable carbocation structural formula of given molecular formula is,
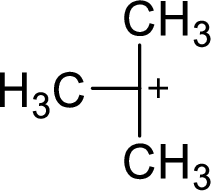
(b)
Interpretation:
The most stable carbocation structural formula of given molecular formula has to be drawn.
Concept Introduction:
The most stable carbocation structural formula:
The most stable structural arrangement of atoms in a carbocation molecule is known as most stable carbocation structural formula.
The highly alkyl substituted carbocation is more stable
Hence, the stability of carbocation is,
(b)
Answer to Problem 9.10P
The most stable carbocation structural formula of given molecular formula is,
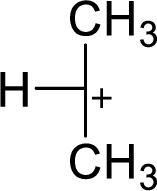
Explanation of Solution
The highly alkyl substituted carbocation is more stable. Hence, the most stable carbocation structural formula of given molecular formula is,
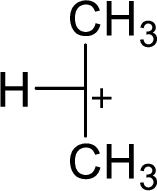
(c)
Interpretation:
The most stable carbocation structural formula of given molecular formula has to be drawn.
Concept Introduction:
The most stable carbocation structural formula:
The most stable structural arrangement of atoms in a carbocation molecule is known as most stable carbocation structural formula.
The highly alkyl substituted carbocation is more stable
Hence, the stability of carbocation is,
(c)
Answer to Problem 9.10P
The most stable carbocation structural formula of given molecular formula is,
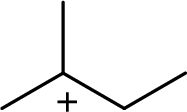
Explanation of Solution
The highly alkyl substituted carbocation is more stable. Hence, the most stable carbocation structural formula of given molecular formula is,
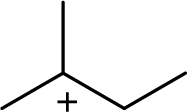
(d)
Interpretation:
The most stable carbocation structural formula of given molecular formula has to be drawn.
Concept Introduction:
The most stable carbocation structural formula:
The most stable structural arrangement of atoms in a carbocation molecule is known as most stable carbocation structural formula.
The highly alkyl substituted carbocation is more stable
Hence, the stability of carbocation is,
(d)
Answer to Problem 9.10P
The most stable carbocation structural formula of given molecular formula is,

Explanation of Solution
The highly alkyl substituted carbocation is more stable. Hence, the most stable carbocation structural formula of given molecular formula is,

Want to see more full solutions like this?
Chapter 9 Solutions
Organic Chemistry
- Which of the two isomers would you expect to undergo E2 elimination faster? trans-1-bromo- 4-tert-butylcyclohexane or cis-1-bromo-4-tert-butylcyclohexane? Draw each molecule in its more stable chair conformation. Provide an explanation to your answer.arrow_forwardDraw the Newman projection of 3-ethyl-2,4-dimethylhexane looking through C3-C4 bond and perform a full conformation alanalysis by rotating along the C2-C3 bond. Make sure to clearly identify the structure of all minima (on the xaxis) and their relative Energies (on they axis).arrow_forwardAccount for the regioselectivity and stereoselectivity observed when 1-methylcyclopentene is treated with reagent. Q) BH3arrow_forward
- Draw the structure of a 1°, 2°, and 3° carbocation, each having molecular formula C4H9+. Rank the three carbocations in order of increasing stability.arrow_forwardArrange the following compounds in the order of increasing stability of carbocations: a. (iv) > (iii) > (ii) > (i) b. (i) > (ii) > (iii) > (iv) c. (i) < (ii) < (iii) < (iv) d. (iv) < (ii) <(i) < (iii)arrow_forwardTreatment of compound A (C8H17Br) with NaOCH2CH3 affords two constitutional isomers B and C. Ozonolysis of B affords CH2=O and (CH3CH2CH2)2C=O. Ozonolysis of C affords CH3CH2CH2COCH3 and CH3CH2CHO. What is the structure of A?arrow_forward
- An optically active unknown compound B, whose molecular formula is C6H10, reacts with H2/Ni produce compound C, whose molecular formula is C6H14. Compound C is optically inactive. What are the structures of compound B and C ?arrow_forwardDehydrohalogenation of 1-chloro-1-methylcyclopropane affords two alkenes (A and B) as products. Explain why A is the major product despite the fact that it contains the less substituted double bond.arrow_forwardDraw the structure consistent with following description. (3E,5Z)-nona-3,5-diene in the s-cis conformationarrow_forward
- The spirocyclic pentadiene derivative F shown below is converted stereospecificallyinto compound G on heating. The transformation involves two consecutive pericyclicreactions of the same type, and proceeds via compound H which is not isolated. Identify the type of pericyclic reaction occurring, and determine the structure ofcompound H.arrow_forwardWrite the structure of the compound E,E-2,4-Hexadien-1-ol and label each non-equivalent carbon with a letter, A,B,C..arrow_forwardArrange the following group in order of increasing priority. Q) -OCH3 -CH(CH3)2 -B(CH2CH3)2 -Harrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
