
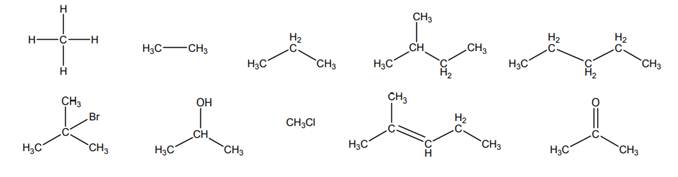
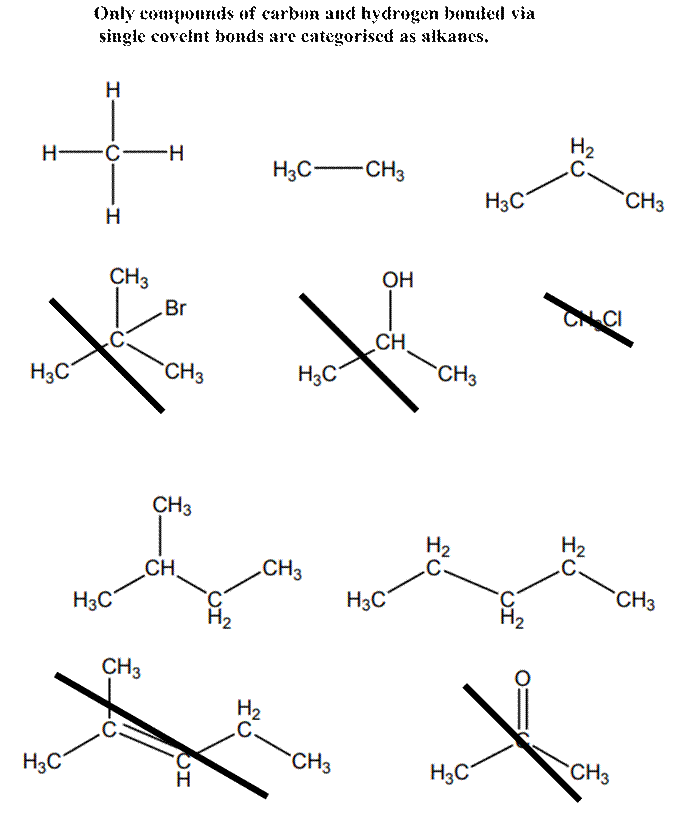
Interpretation: Molecules given below that does not represent an alkane should be crossed out.
Concept introduction:
Explanation of Solution
Alkanes are essentially pure hydrocarbons with no additional
Want to see more full solutions like this?
Chapter NW1 Solutions
Organic Chemistry: A Guided Inquiry
- (E) Write a correct name below each of the unbranched alkanes in CTQ 1.arrow_forwardUsing your model of butane (CH3CH2CH2CH3) , complete the following graph of the anglebetween the two Me groups vs. potential energy. a. Label each Newman projection of butane on the graph with the words staggered, eclipsed, gauche, and anti, as appropriate. (Note that some structures will have more than one label.) b. Draw a wedge and dash bond representation of butane in its lowest P.E. conformation.arrow_forwardA student names the second structure above 2,3-dimethylcyclohex-1-ene. What rule does thisviolate?arrow_forward
- Question: How many C-types do each of the following compounds have? Label them a, b,c ... And Circle the carbon type that would be the most downfield. Please answer all 4 parts of the question. Thank you!arrow_forwardpls help me. The molecule name is vinyl acetatearrow_forwardPlease help wiith the fourth one (C3H8O)arrow_forward
- I'm trying to identify an unknown alcohol. Does this IR belong to a primary, secondary, or tertiary alcohol?arrow_forwardThen please draw and name isomer E if you can please and thank you!arrow_forwardWhich one of the structures in Figure 2 is a tertiary haloalkane? * A B C D None of these compounds are tertiary haloalkanes.arrow_forward
- Please help with the highlighted molecules (i.e. 1 and 2)arrow_forwardBuild a model of 2,2,5,5-tetramethylhexane. Orient the model so that you are looking at the carbon with the arrow pointing to it in Figure 3. Align the bond to the next carbon in the chain so that it is directly behind the first carbon to match a Newman projection view. (See Figure MM.3 in the lab manual) Spin the carbons on either side of the bond you're looking down to cycle through all three staggered and all three eclipsed positions of the substituents. Draw all six positions as Newman projections on the data sheet and identify the position with the highest energy. Draw the six Newman projections of all of the different energy levels. Label each as staggered or eclipsed and rank in order from lowest energy to highest.arrow_forwardI'm looking for an alkene with a single methyl group and an anti markovnikov Br at the last carbon and the markovnikov with Br on the bottom. The image is attached is just to illustrate positioning of the Br, and not an actual alkenearrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning

