3. Provide mechanisms to explain the following reactions. a. H₂O NaOH H₂O H3O 1-iodobutane H3O+ H₂O O based on you mechanistic insight, predict which enantiomer of the product forms HO d. HO NaOEt C4H9 in addition to the mechanism predict which product is favored EtOH e. LDA CH3CO2C(CH3)3 -78 °C
3. Provide mechanisms to explain the following reactions. a. H₂O NaOH H₂O H3O 1-iodobutane H3O+ H₂O O based on you mechanistic insight, predict which enantiomer of the product forms HO d. HO NaOEt C4H9 in addition to the mechanism predict which product is favored EtOH e. LDA CH3CO2C(CH3)3 -78 °C
Chapter30: Orbitals And Organic Chemistry: Pericyclic Reactions
Section30.SE: Something Extra
Problem 41AP: In light of your answer to Problem 30-40, explain why a mixture of products occurs in the following...
Related questions
Question
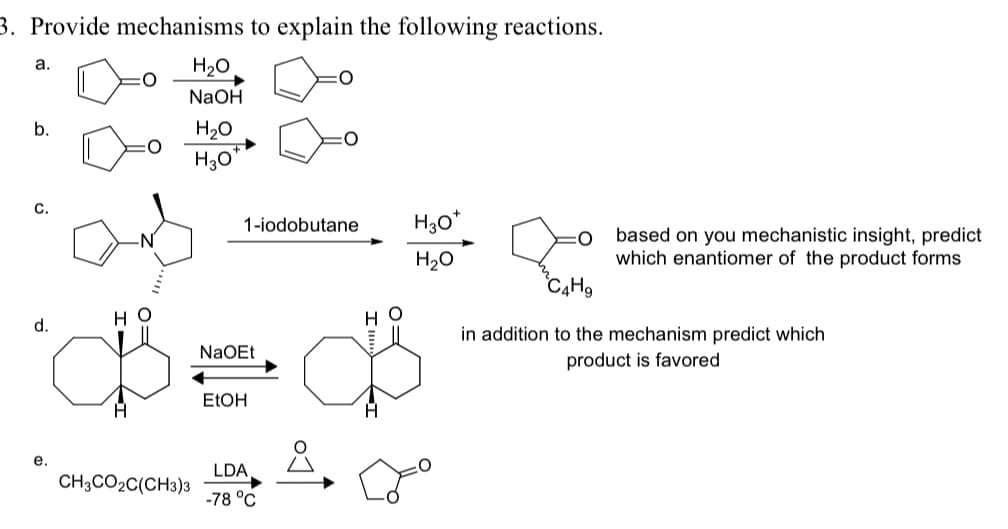
Transcribed Image Text:3. Provide mechanisms to explain the following reactions.
a.
H₂O
NaOH
H₂O
H3O
1-iodobutane
H3O+
H₂O
O
based on you mechanistic insight, predict
which enantiomer of the product forms
HO
d.
HO
NaOEt
C4H9
in addition to the mechanism predict which
product is favored
EtOH
e.
LDA
CH3CO2C(CH3)3
-78 °C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

Recommended textbooks for you

