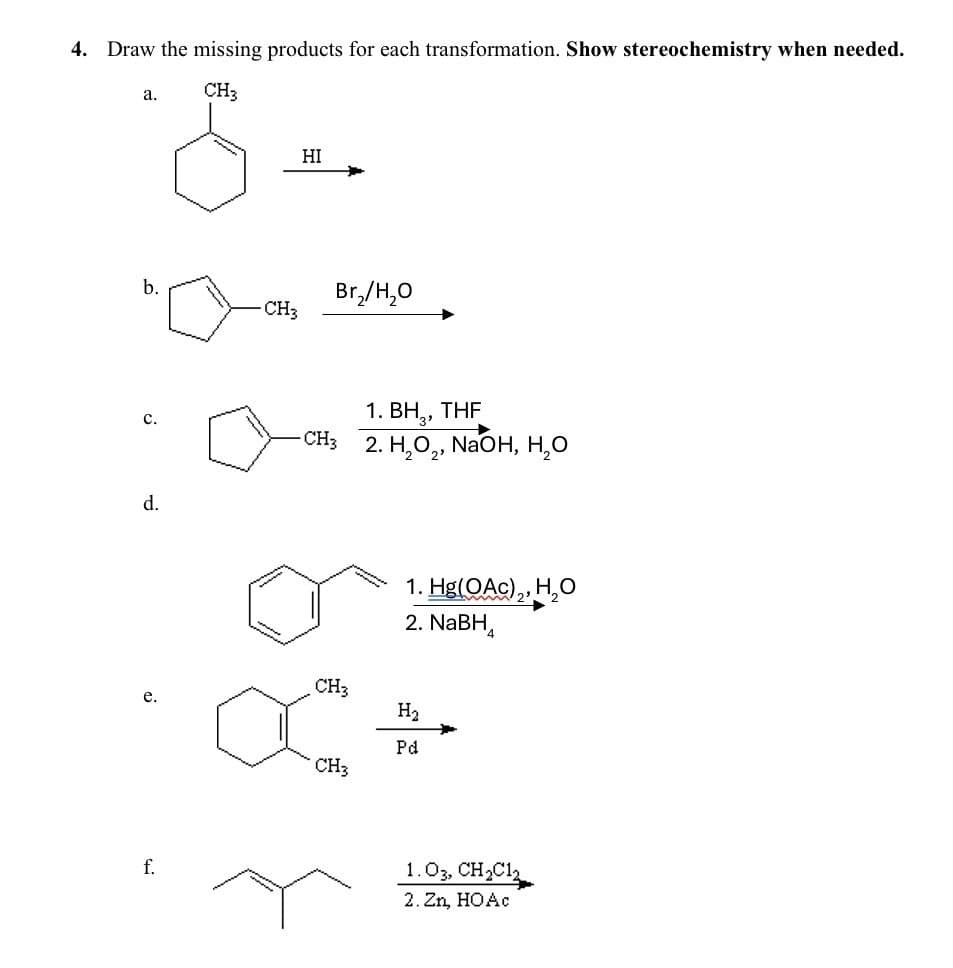
4. Draw the missing products for each transformation. Show stereochemistry when needed. a. CH3 S b. C. d. e. f. HI Br₂/H₂O CH3 1. BH₂, THF -CH3 2. H₂O2, NaOH, H₂O CH3 1. Hg(OAc)2, H₂O 2. NaBH H₂ CH3 Pd 1.03, CH2Cl2 2.Zn, HOẶC
Q: Determine the value of Ksp for Mg(CN), by constructing an ICE table, writing 2 the solubility…
A: Step 1: Solubility product constant (Ksp)Measure of the solubility of a compound in a solvent at a…
Q: Identify the major product. A B AICI 3 ہے C D
A:
Q: Oly H Use the given starting material to synthesize the target molecule. Aim at methods/reagents…
A: The objective of this question is to synthesize a target molecule with an iodine atom from a…
Q: For each of the following compounds, decide whether the compound's solubility in aqueous solution…
A: AgCN: Silver cyanide (AgCN) is sparingly soluble in water. However, its solubility increases with…
Q: Construct the expression for Ksp for solid ZnCO 2 in aqueous solution. 3 ZnCO₂(s) Zn2+(aq) + CO2…
A:
Q: What is the molarity, M, of ions in a 0.40 M aqueous solution of sodium chloride, NaC1? 0.40 M 2.0 M…
A: The objective of this question is to determine the molarity of ions in a 0.40 M aqueous solution of…
Q: O Chemical Reactions = Calculating molarity using solute moles 3/5 Bisho A chemist prepares a…
A: Step 1:- To calculate the concentration of the barium acetate solution, we use the…
Q: Be sure to answer all parts. For the decomposition of gaseous dinitrogen pentaoxide, 2 N2O5(g) → 4…
A: Given: k=2.8x10−3s−1;[N2O5]0=2.31mol/L;t=5.00minStep 1: Write the integrated rate…
Q: If 7.94 g of CuNO3 is dissolved in water to make a 0.860 M solution, what is the volume of the…
A: The objective of this question is to find the volume of the solution in milliliters when 7.94 g of…
Q: 1. If forming new bonds releases energy, how can a reaction ever be endothermic?
A: While it's true that forming new bonds typically releases energy, making a reaction exothermic,…
Q: please help me with my hw. Please answer letter b completely. Hoping for you to help me as i am…
A: The solids are one of the three states of matter and the presence of constituent particles in close…
Q: 15 g of polyethylene glycol is added to 5.0 g of glucose in a bag and then water is added to…
A: Mass of polyethylene glycol = 15 gMass of glucose = 5.0 gInitial volume - 50 mLGlucose Polyethylene…
Q: 0.9583 g of sample was dissolved in 80 mL of xylene and extracted with three 15 mL quantities of…
A: The objective of the question is to calculate the free chloramphenicol content of the sample in…
Q: help 6?
A:
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the carboxylic acid side…
A: Step 1 : Reaction mechanism Step 2 : The answer is :
Q: What are the symmetry elements and point group of each of the following?
A:
Q: Give proper explanation please.
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: O Chemical Reactions Calculating molarity using solute moles 1/5 A chemist prepares a solution of…
A: Given: μmolZn(NO3)2=5.0μmol;V=50.mLC=???μmol/LStep 1: Convert the volume to…
Q: Given Steps step solution
A: Protonation occurs in the first step and by this electrophilicity of the electrophilic centre…
Q: The good expert Hand written solution is not allowed.
A: Note: The left molecule in the image is phenol (C₆H₅OH).The right molecule is ethanol (C₂H₅OH).…
Q: None
A:
Q: None
A:
Q: What is the rusting process for Lead Acid Battery and How to prevent rusting? Explain in details.
A: Here's a breakdown of the corrosion process in lead-acid batteries:When a lead-acid battery is…
Q: None
A: Part 2: Explanation:Step 1: Count the total number of valence electrons.- Chlorine (Cl) has 7…
Q: Show reaekson and don't use hend raiting and step by step solutions
A:
Q: 2. Calculate the Packing Factor of the Sodium Chloride structure to know the degree of occupancy of…
A: Edge Length of Unit Cell (a): The face diagonal (d) is calculated by summing the diameters of the…
Q: None
A: Given: molNi=???mol;molCO=4.72molNi(s)+4CO(g)→Ni(CO)4(s)Step 1: Check if the reaction is…
Q: Please make sure answer is correct a lot of controversial answer on this I will trust you and take…
A: The sequence of reactions is: Double elimination from vicinal dihaloalkanes ⟶ alkyne ⟶cis-alkene ⟶…
Q: Predict the major product of the following reaction: OH Na2Cr2O7 H2SO4, H₂O ? ○ There is no…
A: Step 1:The reagent combination Na2Cr2O7, H2SO4, and H2O is commonly known as chromic acid. It is a…
Q: 11. Supply both sets of missing reagents and the intermediate. (6 pts)
A: Step 1:Friedel-Crafts Acylation: Benzene can be acylated using an acyl chloride (such as propanoyl…
Q: A researcher wants to determine the concentration of beta-carotene in carrots. She obtains a 0.661 M…
A: The objective of this question is to determine the concentration of beta-carotene in Dilution C,…
Q: Enter your answer in the provided box. A radioisotope of iodine, 131I, is used to study thyroid…
A:
Q: Calculate the molarity, M, of a 3.50 liter solution containing 5.00 moles of sodium chloride, NaCl.…
A: The objective of this question is to calculate the molarity of a solution. Molarity is a measure of…
Q: None
A: Step 1:Explanation Step 2: Step 3: Step 4:
Q: The triple alcohol below is treated with excess HCl. Because there is more HCl than needed, all…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A:
Q: None
A: Step 1:C3H8 + O2 → CO2 +H2OWe have to balance this reactionStep 2:Balance Carbon Atoms: Start by…
Q: None
A: Step 1:- Given , Volume of H₂SO₄ solution= 25 ml Molarity of H₂SO₄ solution= 0.200 MMolarity of…
Q: 9.5 9.0 8.5 8.0 7.5 7.0 15.70 1.00 0.88 Unknown 1 CDC13 8.018 7.998 7.953 7.933 -7.562 -7.543 7.524…
A: The objective of the question is to find out the correct pair of the ketone and the alcohol.The…
Q: Assessment 8.50 Predict the products you would get when the following alkenes react under the…
A: Step 1: Step 2: Step 3: Step 4:
Q: Which best describes the overall shape of benzophenone? 4 27 O a. It is planar because all of the…
A: The objective of the question is to determine the overall shape of benzophenone based on the…
Q: Which of the following best describes where the amino acids isoleucine and tryptophan are most…
A: 1. The property of hydrophobicityTryptophan and isoleucine are hydrophobic amino acids. Because of…
Q: dont provide handwriting solution .....
A: The image you sent is a multiple-choice question about the shape of the molecule benzophenone. The…
Q: Show reaekson and don't use hend raiting and step by step solutions please
A: Step 1:Step 2:The reaction simply follows the SN2 mechanism which is a most favoured solvent that is…
Q: Sally obtains a standard calibration curve for their assigned food dye by plotting absorbance versus…
A: The objective of the question is to determine the concentration of the food dye in the original…
Q: Assuming that the dipole moment of a molecule is zero in its electronic ground state (S0) and is µ…
A: The objective of the question is to find the the time evolution of the fluorescence spectrum…
Q: When CO2 is cooled to very low temperatures, it forms a solid known as "dry ice." This solid…
A: Step 1:The best description for dry ice, solid carbon dioxide (CO₂), is:b. A molecular…
Q: For the following compunds 1.Draw the three-dimensional structure includinig hydrogens 2.Put a…
A:
Q: Base
A: Te objective of the question is to find out the mechanism of the given above reaction.In the above…
Q: A 3.0 L container of neon gas has a pressure of 1.0 atm. What will the pressure be if the volume is…
A: The objective of this question is to find the new pressure of the neon gas when the volume is…
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

- 6. Identify the best reagent(s) for this reaction. (see attached screenshot). a. H2SO4, HgSO4, H2O b. 1. Disiamylborane, 2. HO–, H2O2 c. K2Cr2O7, H+ d. NaOCl e. H2, Pd1. Which of the following is the strongest nucleophile? Refer to the diagram below with an MeOH, MeSH, etc. 2. Which of the following is true about the stereochemistry of SN1 reaction?a.retention of configuration at the electrophilic centerb.50:50 mixture of retention and inversion of configuration at the electrophilic centerc.inversion of configuration at the electrophilic centerd. slightly more inversion than retention at the electrophilic centerMaking sure to pay attention to stereochemistry, what is the main product for each reaction?
- These reagents can produce ketones with alkynes A. BH3, THF, H2O2 B. KMnO4 C. O3 D. H2SO4, H2O, HgSO4 choices:A,DB,CA,B,CA,B,C,DWhich of the following reagent best accomplish this transformation below? a. BH3, THF, H2O2 b. NaNH2, NH3 c. H2SO4, H2O, HgSO4 d. H2, LindlarWhich compound will undergo solvolysis most rapidly? A. t-hexyl-I B. t-hexyl-OSO2CH3 C. t-hexyl-F D. t-hexyl-OSO2CF3
- Rank the nucleophiles in each group in order of increasing nucleophilicity. a.−OH, −NH2, H2O b.−OH, Br−, F− (polar aprotic solvent) c.H2O, −OH, CH3CO2−For problem 8.17, all of the reactions will be SN2. For each reaction, identify and evaluate each nucleophile (strong? weak? Strong or weak as a base?) Also, evaluate each solvent as polar protic or polar aprotic. I recommend drawing the structure of each solvent. (g) Sodium methanethiolate (NaSCH3) in ethanolCan you match best nucleophile / conditions from the list that will give a successful hydrolysis reaction for each electrophile? (there's one best nucleophile / conditions for each electrophile) <List for Nucleophile / condition> a. LiAlH4; hydronium work-up b. PCC c. NaH d. CrO3 e. HOEt f. H3O+ or OH- g. H2O h. NaBH4;hydronium work-up Electrophile 1. Acid chloride 2. Acetic anhydride 3. Ester 4. Amide I'm asking again on bartleby because pther tutor gave me the right answer because I think I didn't make my question clear enough.
- The missing reagent should include a bromine atom. and the final product should show stereochemistry.For problem 8.17, all of the reactions will be SN2. For each reaction, identify and evaluate each nucleophile (strong? weak? Strong or weak as a base?) Also, evaluate each solvent as polar protic or polar aprotic. I recommend drawing the structure of each solvent.Consider 3-iodo-2,3-dimethylpentane and 3-iodo-2methylpentane. a. which reacts faster in an Sn2 reaction? Explain. b. which reacts faster in an E2 reaction? Explain.

