Q: N2O4(g) and 1.06 atm NO2(g) at 25°C. The equilibrium reaction is given in the equation below. N2O4(g...
A:
Q: . Part A) What mass in grams of solute is needed to prepare 0.830 L of 0.196 M K2Cr2O7? Part B)...
A: Answer of this question :- Molarity :- Number of mole of solute dissolve in 1 litre of solution.
Q: Please calculate the volume in mL of a solution containing 35.5 grams of sodium sulfate with a molar...
A:
Q: In a calorimetry 0.100 moles of NH4NO3(s) disolved in water. when the temperature decreeses 5.8 degr...
A:
Q: 6)Write the overall rate law for the following reaction: include the units for the rate constant A +...
A: We have to predict overall rate law and rate constant unit
Q: Which are pairs of enantiomers? CH3 CH3 H3C CI CI CH3 H3C ICI ČH2CH2CH3 ČH,CH,CH3 ČH2CH2CH3 I II III...
A: Conditions for pairs of Enantiomers: 1.Enantiomers are non-superimposable mirror images of each ot...
Q: Cyclopentene, C5H8, is a colorless liquid at room temperature that is used in the synthesis of plast...
A: Rate constant of a reaction is defined as the rate of reaction when the concentration of each reacta...
Q: Product(s) (1) MeMgCl, Etz0 (solvent) Ph' Me (2) HCl(aq) "work-up"
A: We have to draw the product of the reaction below, including step by step reaction mechanism with cu...
Q: A 42.0 g sample of compound containing only C and H was analyzed. The results showed that the sample...
A:
Q: Balance the equation: N2(g) + O2(g) → N2O5(g)
A: Simply count the nitrogen and oxygen individually and then proceed further.
Q: Does frequency factor vary with time
A: Frequency factor is also known as pre-exponential factor. Which is very important part of Arrhenius ...
Q: A 2.50 mole quantity of NOCl was initially in a 1.50 L reaction chamber at 400°C. After equilibrium ...
A: Concept: Chemical equilibrium: it is a condition in the course of a reversible chemical reaction in ...
Q: Draw the structure of the conjugate acid of acetone (CH3COCH3)
A: Conjugate acid is a compound that is obtained when a compound gains a proton by reacting with some o...
Q: Write the empirical formula of at least four binary ionic compounds that could be formed from the fo...
A:
Q: Next, the chemist measures the volume of the unknown liquid as 1062. cm° and the mass of the unknown...
A: Given :- Mass of liquid = 1.08 kg Volume of liquid = 1062 cm3 To be determined :- Density of li...
Q: Can I know the step-by-step procedure to calculate these values? 0.2 M Tris = 121.1 g/mole 1 mM Mg...
A:
Q: The half-life for P-32, a radionuclide used in DNA sequencing, is 14.28 days. Calculate the percenta...
A: All natural and artificial radioactive decays follows first order kinetics The integrated rate equat...
Q: According to the following reaction, what volume of 0.365 M Al(NO3)3 is needed to react with 30.0 mL...
A:
Q: Write the empirical formula of at least four binary ionic compounds that could be formed from the fo...
A:
Q: Find all the resonance structures and identify the best contributor
A: We have to draw resonance structure and predict best contributor resonance structure
Q: Buffers are aqueous solutions that tend to resist changes in pH when small amounts of strong acid (H...
A: #Q.15: A buffer solution is an aqueous solution that contains a weak acid/base and its conjugate bas...
Q: For the following reaction 3 experiments have been run and the data collected is in the following ta...
A:
Q: Benzoic acid, HC7H5O2, and its salts are used as food preservatives. Ka for benzoic acid is 6,3 x 10...
A: When an acid undergoes partial dissociation to form protons and conjugate base while a part of the a...
Q: For O(g) at 4200 K, A,G position is 1.000 mol of 0,(g), find the equilibrium composition at 4200 K a...
A: Given: ΔfGo of O (g) = -26,81 KJ/mol. Moles of O2 taken initially = 1.000 mol. Temperature = 4200 K ...
Q: Determine the frequency and energy for light with a wavelength of 712.4 nm. E = kJ/mol What is the c...
A:
Q: Draw diagrams showing how isopropyl alcohol, ethylene glycol and glycerol can hydrogen bond to thems...
A:
Q: Select all of the correct statements about equilibrium from the choices below. At equilibrium the...
A: Equilibrium reaction is a reaction where reaction rate of both forward and backward are equal and th...
Q: For each of the following ionic compounds find the charge on each atom. For example, inAl2O3the oxyg...
A: Charge on each atom
Q: Fe(C5H;)2 + CgH4(=O)2 + HCl (aq) what is the full chemical equation for this?
A: A question based on redox reaction, which is to be accomplished.
Q: a saturated fatty acid sample has a high boiling point. a triacylglycerol is composed of glycerol an...
A: Composition and function of triacylglycerol
Q: A flask is charged with 0.100 mol of A and allowed to react to form B according to the following hyp...
A: We have to calculate the amount of B after given time and also we have to predict rate of disappeara...
Q: 43 46 please answer both
A:
Q: What is the effect on equilibrium when sodium acetate is added to a solution of acetic acid? CH3COOH...
A: The equilibrium reaction taking place is given as, Given: Sodium acetate i.e. NaCH3COO is added to ...
Q: For the following compounds, state whether it is ionic or covalent. If it is ionic, write the symbol...
A: Ionic compounds are compound consisting of ions. Two-element compounds are usually ionic when one el...
Q: (a) Use valence bond theory to identify the sigma (o) bond(s) and pi (7) bond(s) in this molecule. A...
A: The covalent bond is formed by the overlap of the atomic orbitals. On the basis of the type of orbit...
Q: What type of reaction is the following? Ni(s) + H2SO4(aq) → NISO4(aq) + H2(g) combustion decompositi...
A:
Q: An air parcel at temperature 0°C and pressure 70 kPa has a mixing ratio of 4.62 g vapour/kg dry air....
A: Mixing ratio at saturation can be determined in chart. Specific humidity =mass of water vapor per un...
Q: Roviee Topics The concentrations of pollutants are often reported as parts per million (ppm). Parts ...
A: Given: Limit concentration for barium is 2.0 ppm.
Q: Consider the reaction of hydrogen sulfide with methane, given below: 1 CH4(g) + 2 H2S(g) 1 CS2(g) ...
A: "Since you have asked multiple questions, we will solve the first question for you. If you want any ...
Q: H. 2) What is the molecular formula for the molecule shown? OH :NIH Hー HI エ
A: The general molecular formula of any alcohol is - CnHmO where, n and m are the number of carbon and ...
Q: Write the balanced chemical equation that shows the disassociation of the ions and aqueous solution ...
A: Strong acid or strong bases dissociates completely while the weak acid and weak bases doesn't dissoc...
Q: The half-life for gallium-67, a radioisotope, is 78.25 hours. How much of a 56.7 sample of gallium-6...
A: Given datas ; t1/2 = 78.25 hrs. [A]0 = 56.7 mg [A]t = ? t = 359 hrs. t/t1/2 = 359 hrs / 78.25 ...
Q: In an acid-base neutralization reaction, 28.74 mL of 0.500 M potassium hydroxide reacts with 50.00 m...
A: Given: The volume of potassium hydroxide used = 28.74 mL The concentration of the potassium hydroxid...
Q: You just determined that when 14 L of ethane (C2H6) and 14 L of oxygen (O2) combined and burned to c...
A: When ethanol is burnt in presence of O2 , CO2 and H2O are produced as products. Here, in this questi...
Q: Balance the equation: Y2O3(s) + SO3(l) → Y2(SO4)3(s)
A:
Q: Case #2: Ruh-Roh! Old Man Higgins had his favorite cane taken from him when he was out for a walk t...
A: Data analyzed: The CSI team found traces of a pungent liquid at the crime scene. The mass of the sam...
Q: What is the Net ionic equation for the reaction between: K(HC8H4O4)+ NaOH
A: The reactants given are KHC8H4O4 and NaOH.
Q: gallium-67? What are the units for k?
A: Half life time(t1/2) = 78.25 hrs Aa we know Radioactive decay follows first order kinetics Rate c...
Q: The three step mechanism for the following reaction can be seen below ...
A: We have to predict the rate of reaction.
Q: How many carbon atoms does each of the following contain? a) Ba(HCO3)2 b) CH3COOC2H5 c) (CH3)C...
A: a) The compound given is Ba(HCO3)2.
Compare the two highlighted protons and determine which is more acidic. Indicate the factor that was used as your basis.
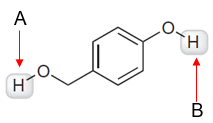
Step by step
Solved in 2 steps with 2 images

- Al+Hg(CH3COO)2Indicate whether hydrogen bonding between like molecules would be important in the following (Yes or no) (a) H2S (b) HF (c) NH3 (d) CO2 (e) CH3OCH3 (f) CH3CH3 (g) CH3OH (h) HBr (i) Cl2 (j) NH4OHExplain why the boiling point of CH3CONH2 (221 °C) is signicantly higher than the boiling point of CH3CO2H (118 °C).





